CBSE Sample Papers for Class 10 Science Paper 1
These Sample papers are part of CBSE Sample Papers for Class 10 Science. Here we have given CBSE Sample Papers for Class 10 Science Paper 1.
Time allowed: 3 hours
Maximum marks : 80
General Instructions
- The question paper comprises two sections, A and B. You are to attempt both the sections.
- All questions are compulsory.
- All questions of Section-A and B are to be attempted separately.
- There is an internal choice in two questions of three marks each and one question of five marks.
- Question numbers 1 and 2 in Section-A are one mark question. They are to be answered in one word or in one sentence.
- Question numbers 3 to 5 in Section-A are two marks questions. These are to be answered in 30 words each.
- Question numbers 6 to 15 in Section-A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section-A are 5 mark questions. These are to be answered in 70 words each.
- Question numbers 22 to 27 in Section-B are based on practical skills. Each question is a two marks question. These are to be answered in brief.
SECTION-A
Question 1.
Define cell respiration. [1]
Question 2.
In which year did the Ganga Action Plan came into being ? [1]
Question 3.
When soap is scrubbed on a stain of curry on a white cloth, why does it become reddish brown, and turns yellow again when the cloth is washed with plenty of water ? [2]
Question 4.
Why is the human eye considered to be special ? [2]
Question 5.
What are the different kinds of consumers seen in a food chain ? [2]
Question 6.
Give reasons for the following: [3]
(a) Why are copper and aluminium wires used as connecting wires ?
(b) Why is lead-tin alloy used for making fuse wires ?
(c) Why is tungsten used for filaments of electric lamps ?
OR
Draw the pattern of magnetic field lines around a current carrying straight conductor. How does the strength of the magnetic field produced change:
(a) With the distance from the conductor ?
(b) With an increase in current in a conductor ?
Question 7.
The far point of a myopic person is 80 cm in front of the eye. What is the nature and power of the lens required to correct the problem? [3]
Question 8.
(a) Name one metal each which can be extracted by: [3]
(i) Reduction with carbon
(ii) Electrolytic reduction
(iii) Reduction with aluminium
(iv) Reduction with heat alone
(b) Give the chemical equation for any two of the above changes.
Question 9.
What is a neutralisation and displacement reaction ? Explain with an example. [3]
Question 10.
How is the opening and closing of the stomata regulated ? [3]
OR
How are the lungs designed in human beings to maximize the area for exchange of gases ?
Question 11.
Name any three glands associated with digestion in humans. Write the names of enzymes secreted by them. [3]
Question 12.
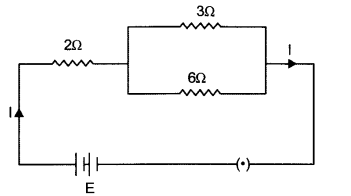
In the given electric circuit if the current flowing through 3 Ω resistor is 1 A, find the voltage of the battery and the current I drawn from it. [3]
Question 13.
What is a homologous series ? [3]
OR
When ethanol reacts with ethanoic acid in the presence of conc. H2SO4, a substance with fruity smell is produced. Answer the following:
(a) State the class of compounds to which the fruity smelling compounds belong. Write the chemical equation for the reaction and write the chemical name of the product formed.
(b) State the role of cone. H2SO4 in this reaction.
Question 14.
Draw a diagram showing the exact location of the pituitary gland, thyroid gland, adrenal gland and pancreas in human beings. [3]
Question 15.
Differentiate between inherited and acquired traits. [3]
Question 16.
Explain the twinkling of stars. What causes it ? [5]
Question 17.
Explain the formation of sodium chloride. Why are they called ionic compound ? Give three point of difference between ionic and covalent compounds. [5]
OR
What are micelles ? What does it form when soap is added to water ? Will a micelle be formed in other solvents such as ethanol also ? State briefly how the formation of micelle help to clean the clothes having oily spots.
Question 18.
Describe the different kinds of fission seen in unicellular organisms with diagram. [5]
Question 19.
Why does a conductor get heated up when an electric current flows through it ? List the factor on which the heat produced in a conductor depends ? State Joule’s law of heating. How will the heat produced in an electric circuit be affected if the resistance in the circuit is doubled for the same current ? [5]
Question 20.

The above two structures have the same formula C4H10. What is the phenomenon and what are they known as ?
Question 21.
(a) State the form in which the following are stored: [5]
(i) Unused carbohydrates in plants.
(ii) The energy derived from food in plants.
(b) Describe the process of nutrition in Amoeba with the help of diagram.
OR
What are the components of the transport system in human beings ? What are the functions of these components ?
SECTION-B
Question 22.
When you add a few drops of acetic acid to a test tube containing sodium bicarbonate powder, which of the following do you observe ? [2]
(a) No reaction takes place.
(b) A colourless gas with a pungent odour is released with brisk effervescence.
(c) Formation of bubbles of a colourless and odourless gas.
(d) A brown coloured gas with brisk-effervescence.
Question 23.
Raju needs hard water to perform an experiment in his laboratory but it is not available on a particular day. There are some salts in the laboratory which when dissolved in water can convert it into hard water. Select which of the following group can be used ? [2]
(a) Sodium chloride and potassium chloride
(b) Sodium sulphate and potassium sulphate
(c) Sodium sulphate and calcium sulphate
(d) Calcium sulphate and calcium chloride.
Question 24.
Show the different stages in binary fission of Amoeba. [2]
Question 25.
A student on observing the embryo of a gram seed listed the various parts he saw-testa, tegmen, cotyledon, micropyle, plumule and radicle. On checking the teacher found only three correct parts. These are as follows: [2]
(a) Cotyledon, testa, plumule
(b) Cotyledon, plumule, radicle
(c) Cotyledon, tegmen, radicle
(d) Cotyledon, micropyle, plumule.
Question 26.
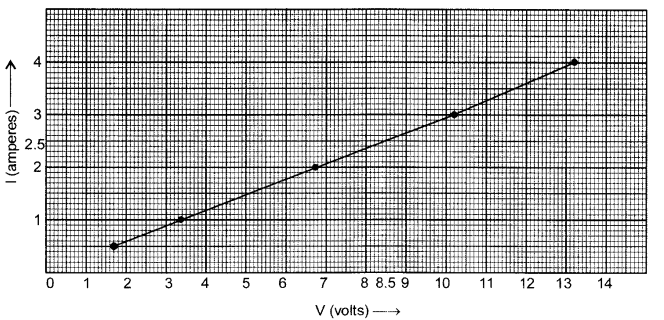
The values of current I flowing in a given resistor for the corresponding values of potential difference V across the resistor are given below: [2]
| I (amperes) | 0.5 | 1.0 | 2.0 | 3.0 | 4.0 |
| V (volts) | 1.6 | 3.4 | 6.7 | 10.2 | 13.2 |
Plot a graph between V and I.
OR
Draw a circuit diagram to show three resistors connected in series with a cell and a key. Write formula for the equivalent resistance R of this combination if the individual resistances of the resistors are R1, R2 and R3.
Question 27.
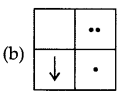
If you focus the image of a distant object whose shape is given below, on a screen using a convex lens. [2]
The shape of the image of this object on the screen would be:

ANSWERS
SECTION-A
Answer 1.
Cell respiration can be defined as the biological oxidation of food taking place partly in the cytoplasm and partly in the mitochondria of the cell. During this oxidation, carbon dioxide and water are formed with the release of energy. This energy is stored in the cell as ATP-Adenosine Tri-Phosphate which is used when required to perform the various activities of the body.
Answer 2.
The Ganga Action Plan came into being in the year 1985. This multi-crore project came into being because the quality of water in the Ganga was very poor. Coliform bacteria, found in human intestine and whose presence indicates contamination by disease causing micro-organisms were found here. There were other dangerous contaminants too.
Answer 3.
The curry might contain turmeric powder which is the indicator of acids and bases. Soap being basic in nature turns the colour of the turmeric from yellow to reddish brown. After washing with lot of water the soap is removed and the turmeric returns of its yellow colour.
Answer 4.
The human eye is the most valuable and sensitive sense organ in our body. It helps us to see the wonderful and colourful world around us. If we close our eyes we can identify objects by their smell, sound, taste and feel. We can identify colours only by seeing them. The eye is like a camera. Its lens forms an image on the light-sensitive screen called retina. The ciliary muscles adjust the focal length of the lens so that wherever the object is placed, the image is always formed on the retina. The pupil regulates and controls the amount of light entering the eye. The precious eyes are protected by being placed in hollows of the skull called orbits.
Answer 5.
In any ecosystem we find several organisms interacting with one another. The process of different types of organisms feeding on one another constitutes a food-chain. Each step of the food chain is called a trophic level. Green plants which trap solar energy and prepare food are called producers. They are eaten by the herbivores which are called primary consumers. These are further eaten by small carnivores which are called secondary consumers. They in turn are eaten by big consumers called tertiary consumers.
Answer 6.
(a) Copper and aluminium wires are used as connecting wires because they have low resistivity and are good conductors of electricity.
(b) Lead-tin alloy is used for making fuse wires because it has low melting point.
(c) Tungsten has high melting point and has great tensile strength so it is used as a filament in electric lamps.
OR
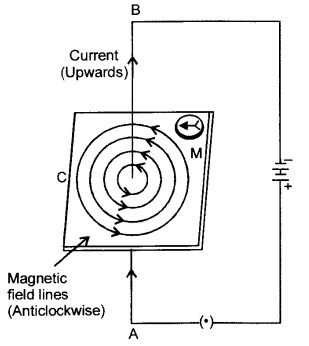 (a) The strength of a magnetic field is inversely proportional to the square of the distance from the conductor i.e., the strength of the electric field decreases with increase in distance.
(a) The strength of a magnetic field is inversely proportional to the square of the distance from the conductor i.e., the strength of the electric field decreases with increase in distance.
(b) The strength of the magnetic field is directly proportional to the current passing in the wire i.e., strength of the magnetic field increases with increase in current.
Answer 7.
The person is suffering from an eye defect called myopia. In this defect, the image is formed in front of the retina. Hence, a concave lens is used to correct this defect of vision.
The object distance (μ) = ∞
The image distance (υ) = – 80 cm = – 0.8 m
Using the formula,
\(\frac { 1 }{ \upsilon } -\frac { 1 }{ u } =\frac { 1 }{ f }\)
Substituting the values, we get
\(\frac { -1 }{ 0.8 } -\frac { 1 }{ \infty } =\frac { 1 }{ f } \)
\(\frac { -1 }{ 0.8 } -0=\frac { 1 }{ f } \quad \)
\(\frac { 1 }{ f } =\frac { -1 }{ 0.8 } =-1.25D \)
A concave lens of power -1.25 D is required by the person to correct his defect.
Answer 8.
(a) (i) Zinc
(ii) Aluminium
(iii) Magnesium
(iv) Mercury
(b) (i) ZnO + C→ Zn + CO
(ii) 3MgO + 2Al→ 3Mg + Al03
Answer 9.
Whenever an acid reacts with a base it results in the formation of a salt and water and this is called neutralisation reaction.
Acid + Base = Salt + Water
NaOH(aq) + HCl(aq)→ NaCl(aq)+ H2O(1)
In the above case when sodium hydroxide reacts with hydrochloric acid they react and release their OH– and H+ ions. These two ions combine to form water. Now sodium and chloride ions combine among themselves and form the salt sodium chloride.
Those reactions in which one element takes the place of another element in a compound, are as displacement reactions. In general, a more reactive element displaces a less reactive element compound. The examples of some important displacement reactions are given
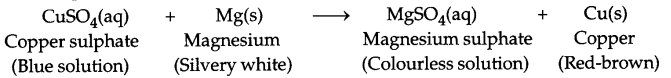
Answer 10.
The opening and closing of stomata is regulated by the activity of the guard cells. Guard cells are 2 kidney shaped cells present on either side of the stomata. They contain chloroplast. The space between the two guard cells is called the stomatal pore. When water flows into the guard cells they swell up thereby opening the stomatal pore. When guard cells lose water they shrink and the stomatal pore closes. Stomata are present on the surface of the leaves. Generally plenty of gaseous exchange takes place through these pores for photosynthesis. As there is a tendency to lose a lot of water through these pores by transpiration, the plant tends to keep the pores closed when gaseous exchange does not take place.
OR
Lungs are elastic, spongy, moist sacs like structures. Gaseous exchange occurs between alveoli and the blood vessels surrounding alveoli. Alveoli are tiny sacs like structures present in lungs which increases the surface area for gaseous exchange and lungs contain about 300-500 million alveoli. They have very thin walls making the diffusion of gases more convenient. They are surrounded by numerous tiny blood capillaries which facilitates for efficient gaseous exchange. During inhalation the ribs moves outward and diaphragm moves downwards so the space inside the thoracic cavity increases letting more amount of oxygen to diffuse inside.
Answer 11.
| S. No. | Glands | Enzymes |
| (i) | Salivary glands | Salivary amylase |
| (ii) | Pancreas |
Pancreatic amylase Trypsin Lipase |
| (iii) | Gastric glands | Pepsin |
Answer 12.
Given: R2 = 3 Ω, I2 = 1 A
V2 = I2 × R2=1 ×3=3V

Answer 13.
A series of compounds in which the same functional group substitutes for hydrogen in a carbon chain is called a homologous series. Carbon atoms have the ability to be linked together to form chains of varying lengths. In addition, hydrogen atoms or atoms on these carbon chains can be replaced by any of the functional groups like alcohol, ketone, aldehyde, carboxylic acid and so on. The presence of the functional group dictates the properties of the carbon compound regardless of the length of the carbon chain. If we consider CH3OH, C2H5OH, C3H7OH, C4H9OH, their chemical properties are similar as all of them contain the OH group. As the molecular mass increases in the homologous series we can see a gradation in physical properties. The melting and boiling points increase with increasing molecular mass. Solubility in a particular solvent also increases. Chemical properties remain the same as they are determined by the functional group.
OR
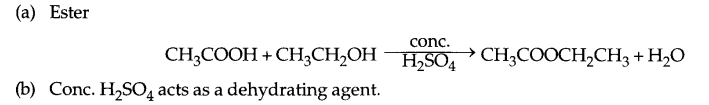
Answer 14.
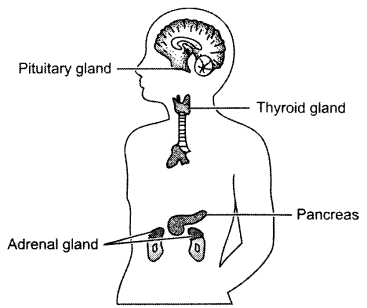
Answer 15.
| Inherited traits | Inherited traits |
| (i) These are traits controlled by the DNA. | These are traits not controlled by the DNA |
| (ii) These traits are inherited. | These traits cannot be inherited. |
| (iii) These traits are not affected by the environment. Experience of the individual during its lifetime | These traits are due to the environment or |
| (iv) These traits can lead to variation which combined with genetic drift can lead to speciation. | These traits do not lead to variation and do not play any major role in evolution. |
| (v) Example: Attached or free earlobe and curly hair body. | Example: Learning dance, music, muscular of a wrestler. |
Answer 16.
Sometimes we can see a random wavering or flickering of objects through a turbulent stream of hot air rising above a fire or a radiator. The air above the fire turns hotter than the air further up. The hotter air is lighter than the cooler air above it and the refractive index is also lesser than that of the cooler air. As the physical condition of the refracting medium (air) are not stationary, the apparent position of the object as seen through hot air fluctuates. This wavering is an effect of atmospheric refraction on a small scale in our local environment. The twinkling of stars is an effect of atmospheric refraction on a much larger scale. The starlight, on entering the earth’s atmosphere undergoes refraction continuously before reaching the earth.
Since the atmosphere bends starlight towards the normal, the apparent position of the star is slightly different from its actual position. The star appears higher than its actual position when viewed near the horizon. Moreover the position of the star keeps changing as the physical conditions of the earth’s atmosphere are not the same. As the stars are far away, the path of the light rays coming from them keeps varying slightly and the starlight entering the eye flickers. The star appears brighter at times and fainter at times, this produces the twinkling effect.
Answer 17.
Sodium chloride is formed by the combination of sodium and chloride. Sodium has the electronic configuration 2,8,1. Chloride has the electronic configuration 2,8,7. When both of them combine, sodium tends to lose one electron to chloride and become \({ Na }^{ + } \) (sodium cation). Chloride picks up that electron and becomes \({ Cl }^{ – } \) (chloride anion). Now sodium (\({ Na }^{ + } \)) gets the stable configuration of 2, 8 and chloride (\({ Cl }^{ – } \)) gets the stable configuration 2,8,8. Sodium and chloride ions, being oppositely charged, attract each other and are held together by strong electrostatic forces of attraction to exist as sodium chloride (NaCl). As the compound sodium chloride is formed by the combination of sodium and chloride ions, the oppositely charged ions.
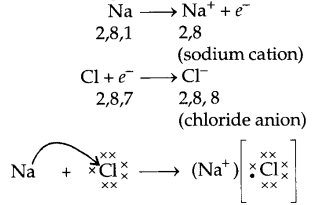
|
Ionic compounds |
Covalent compounds |
| (i) They are formed by the transfer of electrons that are positively and | They are formed by the sharing of the electrons. |
| (ii) They have high melting and Boiling | They have low melting and Boiling points. |
| (iii) They are soluble in water. | They are insoluble in water. |
OR
A soap molecule has two parts; one is non-polar, hydrophobic and other is polar-hydrophilic. When soap is added to it, the polar ends dissolve in water while the non-polar end dissolve in each other. As a result, micelle formation takes place. Since soap is soluble in ethanol, micelle formation does not take place. The micelle formation takes place because their hydrocarbon chains come together and the polar ends are projected outwards. The dirt present on clothes is organic in nature and insoluble in water. Therefore, it cannot be removed by only washing with water. When soap is dissolved in water, its hydrophobic ends attach themselves to the dirt and remove it from the cloth. Then, the molecules of soap arrange themselves in micelle formation and trap the dirt at the centre of the cluster. These micelles remain suspended in the water. Hence, the dust particles are easily rinsed away by water.
Answer 18.
Fission or cell division leads to the creation of new individuals in unicellular organisms. There are different patterns of fission. Many bacteria and protozoa just split into two equal halves during cell division. In Amoeba, fission can take place in any plane.
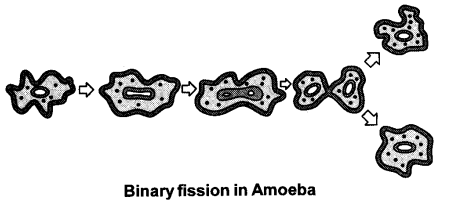
Other unicellular organisms like Leishmania show binary fission along a definite plane. Leishmania is a protozoan that causes the disease kala-azar. It has a better organisation in its body and has a long whip like flagellum at one end.
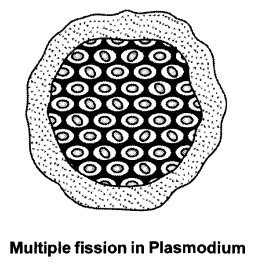
Other unicellular organisms like the malarial parasite, Plasmodium divide into several daughter cells at the same time by the process of multiple fission.
Another unicellular organism like yeast develops a large number of buds which separate from the parent body by fission. These buds then start growing further into new individuals.
Answer 19.
When an electric current is passed through a conductor it gets heated. This is the heating effect of current. It is obtained by the transformation of electrical energy into heat energy. A battery of a cell is the source of electrical energy. The chemical reaction within the cell generates the potential difference between its two terminals that sets the electrons in motion to make the current flow through a resistor. The source has to keep expending its energy. A part of the source energy in maintaining the current is consumed into useful work and the rest of it is expended as heat.
The heat produced in a wire is directly proportional to:
- The square of the current (\({ I }^{ 2 } \)).
- Resistance of wire (R).
- Time (t) for which the current passes.
Joule’s law states that when a current of I amperes flows in a wire of resistance of R ohms for time t seconds, the heat produced in the conductor is equal to the product of the square of the current, resistance of the wire and time for which the current is passed.
By Joule’s law of heating, H = \({ I }^{ 2 } \) x R x t
As H ∝ R, therefore if the resistance in the circuit is doubled, heat produced will also double.
Answer 20.
The given two structures have the same formula C4H10. However their structures are different. Compounds having the same molecular formula but different structural formula are called structural isomers. All carbon compounds which have just carbon and hydrogen in them are called hydrocarbons. Compounds of carbon which have single bonds between the carbon atoms are called saturated hydrocarbons. They are known as alkanes. The other compounds of carbon which have double or triple bonds between the carbon atoms are called unsaturated hydrocarbons. They include, alkenes and alkynes
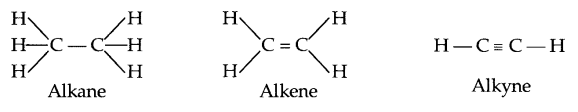
Answer 21.
(a)
- Unused carbohydrates are stored as starch in plants.
- The energy derived from food is stored in the form of ADP-Adenosine Di-Phosphate and ATP- Adenosine Tri-phosphate.
(b) Nutrition in Amoeba : Amoeba is the simplest form of unicellular animals. It consumes food using its pseudopodia or false feet. They are temporary finger-like projections which can develop at any time. They grow and fuse around the food particle. This results in the formation of the food vacuole. Inside the food vacuole the food particle is digested with the help of enzymes and broken down into simpler substances. These are then absorbed into the cytoplasm. The remaining unwanted material is sent out of the cell.
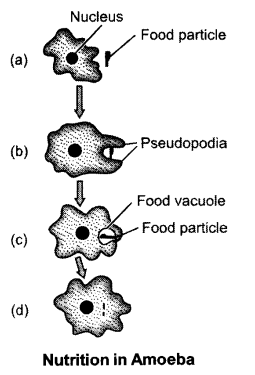
OR
The components of the transport system in human beings are :
- Heart: It is the central pumping organ which pumps blood to various parts of the body.
- Blood : It is a mobile, red coloured fluid that circulates within the blood vessels in the body. It supplies nutrients and oxygen to all the living cells and removes waste products and carbon dioxide from them.
- Blood vessels: They are elastic muscular tubes which carry blood. There are three kinds of blood vessels:
(a) Arteries : They carry oxygenated blood from heart to different parts of the body except pulmonary artery.
(b) Veins : They carry de-oxygenated blood to heart from different parts of the body except pulmonary vein.
(c) Capillaries : They are thin walled, narrow blood vessels which connect arteries and veins. Through capillaries exchange of gases, diffusion of nutrients, waste products etc. occurs between body cells and blood. - Lymph : It carries digested and absorbed fat from intestine to other tissues as well as drains excess fluid from extracellular space back into the blood.
SECTION-B
Answer 22.
(c) Formation of bubbles of a colourless and odourless gas.
Answer 23.
(d) Calcium sulphate and calcium chloride.
Answer 24.
Binary fission means the splitting of the parent cell into two cells. It is a kind of asexual reproduction in Amoeba. The two equal sized cells are called off-springs and they have a copy of their parent’s generic material.
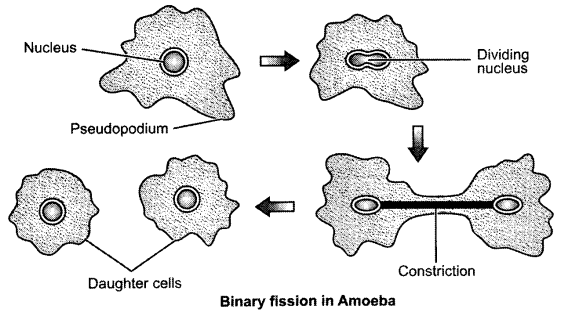
Answer 25.
(c) Cotyledon, tegmen, radicle.
Answer 26.
The graph between V and I plotted by using the given data is shown below:
OR
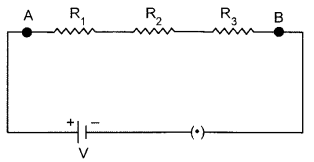
If Rs is the equivalent resistance of the given parallel combination, then
RS = R1 + R2 + R3
Answer 27.
We hope the CBSE Sample Papers for Class 10 Science paper 1 help you. If you have any query regarding CBSE Sample Papers for Class 10 Science paper 1, drop a comment below and we will get back to you at the earliest.
