CBSE Sample Papers for Class 10 Science Paper 3
These Sample papers are part of CBSE Sample Papers for Class 10 Science. Here we have given CBSE Sample Papers for Class 10 Science Paper 3
Time allowed: 3 hours
Maximum marks : 80
General Instructions
- The question paper comprises two sections, A and B. You are to attempt both the sections.
- All questions are compulsory.
- All questions of Section-A and B are to be attempted separately.
- There is an internal choice in two questions of three marks each and one question of five marks.
- Question numbers 1 and 2 in Section-A are one mark question. They are to be answered in one word or in one sentence.
- Question numbers 3 to 5 in Section-A are two marks questions. These are to be answered in 30 words each.
- Question numbers 6 to 15 in Section-A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section-A are 5 mark questions. These are to be answered in 70 words each.
- Question numbers 22 to 27 in Section-B are based on practical skills. Each question is a two marks question. These are to be answered in brief.
SECTION-A
Question 1.
Name one flower that has both stamen and pistil. [1]
Question 2.
Which enzyme is present in saliva and what is its function ? [1]
Question 3.
What is metallic lustre ? How are metals found in nature ? What is gangue ? [2]
Question 4.
What is accommodation of the eye ? When does the maximum accommodation of a normal eye reached. [2]
Question 5.
“Burning of fossil fuels results in global warming.” Give reasons to justify this statement. [2]
Question 6.
Define absolute and relative refractive index ? [3]
OR
Draw ray diagrams to show the formation of three times magnified (a) real, and (b) virtual image of an object by a convex lens. Mark the positions of O, F, 2F in the diagrams.
Question 7.
An electric oven of 2 kW power rating is operated in a domestic electric circuit (220 V) that has
a current rating of 5 A. What results do you expect ? Explain. [3]
Question 8.
What are the indicators that suggest that a chemical reaction has taken place ? Give two examples of combination reaction. [3]
Question 9.
What happens when a magnesium ribbon is burnt in air ? What kind of change is it ? Why magnesium ribbon be cleaned before burning in air ? [3]
Question 10.
What are homologous organs ? What do they point to ? Give an example. [3]
OR
What are the different modes of reproduction seen in unicellular organisms ? Give examples.
Question 11.
Compare anaerobic and aerobic respiration. [3]
Question 12.
(a) What is a coulomb ? [3]
(b) Define electric circuit. Give the circuit diagram of a typical electrical circuit.
Question 13.
Why should a chemical equation be balanced ? Balance the following equations: [3]
(a) HNO3 + Ca(OH)2 → Ca(NO3)2 + H2O
(b) NaCl + AgNO3 → AgCl + NaNO3
OR
Why is respiration considered an exothermic reaction ? Explain.
Question 14.
What was Chipko Andolan ? How did this Andolan ultimately benefit the local people and the environment ? [3]
Question 15.
How is food digested in the small intestine ? What are the different substances that act upon and break down carbohydrates, proteins and fats. [3]
Question 16.
It is desired to obtain an erect image of an object, using concave mirror of focal
length of 12 cm. [5]
- What should be the range of distance of an object placed in front of the mirror ?
- Will the image be smaller or larger than the object ? Draw ray diagram to show the formation of image in this case.
- Where will the image of this object be, if it is placed 24 cm in front of the mirror ? Draw ray diagram for this situation also to justify your answer.
Show the positions of pole, principal focus and the centre of curvature in the above ray diagrams.
Question 17.
Both soap and detergent are some type of salts. What is difference between them ? Describe in brief the cleansing action of soap. Why do soaps not form lather in hard water ? List two problems that arise due to the use of detergents instead of soaps. [5]
OR
- How can we convert unsaturated hydrocarbon into saturated hydrocarbon ?
- How would you distinguish experimentally between an alcohol and a carboxylic acid on the basis of a chemical property ?
Question 18.
Draw a neat labelled diagram of the human brain. Write down the functions of the cerebellum and medulla oblongata ? [5]
Question 19.
- How is the strength of the magnetic field at a point near a straight conductor related to the strength of the electric current flowing in the conductor ? [5]
- With the help of a diagram describe an activity to show that a straight conductor carrying current produces a magnetic field around it. State the rule which maybe used to determine the direction of magnetic field thus produced.
- Why do two magnetic field lines never intersect each other ?
Question 20.
What is the major difference between Mendeleev’s periodic table and the Modem Periodic Table ? Discuss the trend in the modem periodic table with regard to valency and atomic size. [5]
Question 21.
What is a good source of energy ? Name some non-conventional sources of energy that man is trying to use now-a-days. What will be the environmental consequence of these energy
sources that man needs to be careful about ? [5]
OR
- What are fossil fuels and why are they called so ?
- How are they important for us ?
- Name two fossil fuels ?
- Give some methods to conserve fossil fuels ?
SECTION-B
Question 22.
When water is slowly added to calcium oxide or quick lime in a beaker the solution turns hot. Why ? [2]
Question 23.
Why are tarnished copper vessels washed in lemon or tamarind juice ? [2]
Question 24.
A student was asked to observe the permanent slide of yeast showing asexual reproduction. What was the process he saw ? Let him draw the diagram. [2]
Question 25.
Name the type of asexual reproduction in which two individuals are formed from a single parent and the parental identity is lost. Write the first step from where such a type of reproduction begins. Draw first two stages of this reproduction. [2]
Question 26.
Tina got her eyes tested and doctor recommended -3D lens for correcting her vision. [2]
- What kind of eye defect is Tina suffering from ?
- What could be the cause of this defect ?
OR
Draw a ray diagram to show the formation of image by a convex lens when an object is placed in front of the lens between its optical centre and principal focus.
Question 27.
If a student wants to connect four cells of 1.5 V each to form a battery of voltage 6 V, then how would he draw the symbol of the battery ? [2]
ANSWERS
SECTION-A
Answer 1.
Hibiscus plant has both stamen and pistil.
Answer 2.
Salivary amylase is present in saliva and its function is to break down starch into sugar.
Answer 3.
Metallic lustre refers to the shining appearance of metals in their pure state. Metals are usually found in the form of minerals in nature. A mineral contains a high percentage of the metal. These minerals are called ores. Ores mined from the earth contain a lot of impurities like soil, sand etc. called gangue.
Answer 4.
The ability of an eye to focus the distant objects as well as the nearby objects on the retina by changing the focal length of its lens is called accommodation. The maximum accommodation of a normal eye is reached when the object is at a distance of about 25 cm from the eye.
Answer 5.
Burning of fossil fuels produces green house gases like CO, CO2 etc. High concentration of these gases in the atmosphere causes insulation, creates green house effect and causes global warming.
Answer 6.
Absolute refractive index: It is the refractive index of the medium defined with respect to a vacuum. It is the ratio of the speed of light in vacuum to the speed of light in the medium. It is always greater than one.
Relative refractive index: It is the refractive index of the medium defined relative to another medium. It is the ratio of speed of light in one medium (other than vacuum or air) to the speed of light in another medium
OR
(a) To get three times magnified real image,the object has to be placed between F1 and 2F1
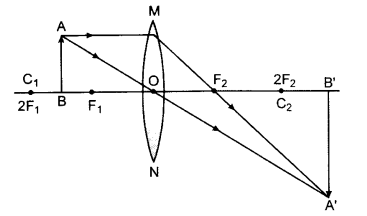
(b) To get three times magnified virtual image, the object is placed between F1 and the optical centre O.
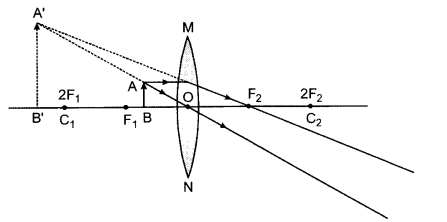
Answer 7.
An electric oven of 2 kW rating means it has a total power of 2 x 1000 = 2000 W. Now this is operated in a domestic circuit of 220 V. Hence, the current flowing will be 2000 W/220 V. This means that the 1 current drawn by the electric oven is 9.09 A which is very high but the fuse in this circuit is only of 5 A capacity. So, when a very high current of 9.09 A flows through the 5 A fuse, the fuse wire will get heated too much, melt and break the circuit, cutting off the power supply.
Answer 8.
The indicators that suggest that a chemical reaction has taken place are as follows :
- Change in state,
- Change in colour,
- Change in temperature,
- Evolution of a gas,
- Formation of precipitate.
Examples of combination reaction are:

Answer 9.
When magnesium ribbon is burnt in air, it bums with a dazzling white flame. It undergoes oxidation and forms magnesium oxide. It is a chemical change irreversible in nature.
It is cleaned before burning in air in order to remove the protective layer of basic magnesium carbon¬ate from its surface so that it may readily combine with oxygen present in the air to form magnesium oxide.
![]()
Answer 10.
Homologous organs are the organs having the same structure but different functions. They point to a common ancestor. They help to identify an evolutionary relationship between apparently different species. Some examples are the forelimb of a frog, the forelimb of a lizard, the wing of a bird and the hand of humans. However all similarities in organ shape does not imply a common ancestry. If we consider the wings of bats and birds we find they have the same function but different structure. The wing of bat is made up of a fold of skin, the wing of birds is made up of feathers. This means they are not homologous but analogous.
OR
The different modes of reproduction seen in unicellular organisms are fission and budding.
In Amoeba, cell division leads to the formation of two new individuals. When two individuals are formed it is called binary fission. Binary fission is also seen in Leishmania, When many individuals are formed as in Plasmodium it is called multiple fission. In yeast, many small buds are formed in the parent body which separate and form new individuals.
Answer 11.
| Anaerobic respiration | Aerobic respiration |
| (i) It takes place in the absence of oxygen. | It takes place in the presence of oxygen. |
| (ii) It usually takes place in fungi during fermentation. | It takes place in all plants and animals. |
| (iii) Glucose is broken down into ethyl alcohol and carbon dioxide. | Glucose is completely oxidised to form carbon dioxide and water. |
| (iv) Small amount of energy is released here. | Plenty of energy is released which is stored as ATP-Adenosine Tri Phosphate molecules. |
| (v) The process is often toxic to both plants and animals. | The process is non-toxic to both plants and animals |
Answer 12.
(a) Coulomb is the S.I. unit of electric charge. It is equivalent to the charge contained in
6 x 1018 electrons.
(b) A continuous conducting path consisting of wires and other resistances (like electric bulb, etc.) and a switch, between the two terminals of a cell or a battery along which an electric current flows, is called an electric circuit.
The circuit diagram of a typical electrical circuit is given below:
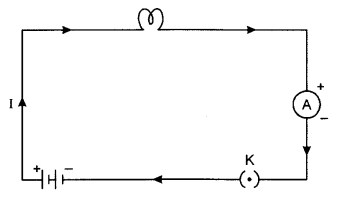
Answer 13.
As per the law of conservation of mass, mass can neither be created nor be destroyed in a chemical reaction. So the total mass of the elements present in the products of a reaction has to be equal to the total mass of the elements in the reactants. This means the number of atoms of each element remains the same before and after a chemical reaction.
- 2HNO3 + Ca(OH)2 → Ca(NO3)2 + 2H2O
- NaCl + AgNO3 → AgCl + NaNO3
OR
In human beings, energy is obtained from the food we eat. During digestion of food, large molecules of food like carbohydrates, proteins etc. are broken down into simpler substances such as glucose, amino acids etc. Glucose combines with oxygen in the cells and provides energy. The special name of this combustion reaction, Since energy is released in the whole process, it is an exothermic process.
Answer 14.
Chipko movement means “hug the tree” it is one of the movements in India to conserve biodiversity. It was started by Sunder Lai Bahuguna. This movement originated in Reni Village of Garhwal. To stop the contractor from falling of trees women in the village clasped to the trunk of the tree. Its benefits were:
- Existing forest cover was protected, reducing landslides and soil erosion. It actually protected environment
and maintained ecological balance. - Forest wealth could be utilized for food, fodder, fuel, fertilizers and fibres.
Answer 15.
The small intestine is the part where complete digestion of carbohydrates, proteins and fats takes place. The secretions of the liver and pancreas reach the small intestine. The digested food from the stomach is acidic and it mixes with the bile juice from the liver to change to alkaline medium for the pancreatic enzymes to act on it. Bile salts break down the big fat globules into smaller parts for further digestion. Pancreatic juice has enzymes like trypsin to act on proteins and lipase to act on emulsified fats. The walls of the small intestine have glands that secrete the intestinal juice. They convert protein to amino acids, carbohydrates to glucose and fats into fatty acids and glycerol.
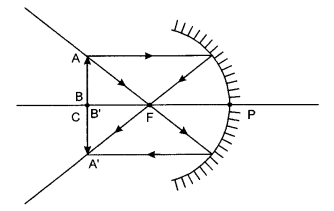
Answer 16.
(a) Range of distance should be 0 cm to < 12 cm.
(b) The image will be larger than the object.
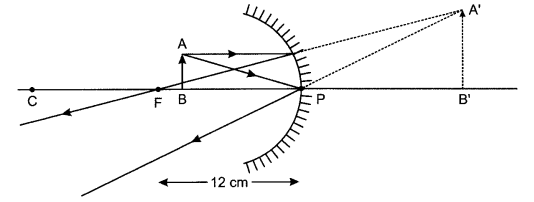
(c) Image will be formed at a distance of 24 cm in front of the mirror
Answer 17.
Soaps are made up of sodium or potassium salts of long chain carboxyic acids.
Detergents are sodium salts of bezene sulphonic acid.
Cleansing action of soaps :
One part of soap is ionic, it is hdrophilic and dissolves in water. The other part is non-ionic consists of hydrocarbon chain and is hydrophobic in nature which is insoluble in water but dissolves in oil. Thus, soap molecules arrange themselves in the form of a micelle. On rinsing with water, soap is washed off, lifting the oily dirt particles with it. Soap does not form lather in hard water because of the reaction of soap with Ca2+ and Mg2+ ions present in hard water which forms insoluble precipitate called as scum.
Problems due to the use of detergents are:
- Detergents are non-biodegradable.
- It leads to water or air pollution that can also cause the skin problems.
OR
(a) By the addition reaction of unsaturated hydrocarbon with halo atom in the presence of catalyst.
HC = CH + H2 \(\xrightarrow [ 5700K ]{ Ni }\) CH3 – CH3
R2C = CR2 + H2 \(\underrightarrow { Ni } \) R2HC – CHR2 (Vegetable Gee)
This reaction is known as Hydrogenation reaction.
(b) Take alcohol and a carboxylic acid in two separate test tubes. Add sodium hydrogen carbonate in both the test tubes. The test tube, from which the effectiveness evolves, contains carboxlyic acid. The effervesecence is due to the evolution of carbon dioxide gas. However, from test tube containing alcohol there is no effervescence, because no carbon dixode gas is produced there.
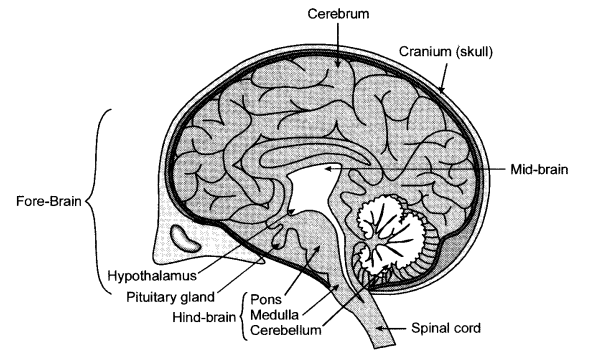
Answer 18.
The cerebellum is a part of the hind-brain and it is responsible for voluntary actions and for main-taining balance and posture of the body. The medulla also a part of the hind-brain controls many involuntary actions like maintaining the blood pressure, salivation and vomiting. Moreover the medulla is the connection between the brain and the spinal cord.
|
Cerebellum |
Medulla oblongata |
| (i) The cerebellum is a part of the hind-brain. | The medulla is also a part of the hind-brain. |
| (ii) It controls voluntary actions of the body like maintaining balance and posture. | The medulla controls involuntary actions of the body like salivation, vomiting and blood pressure. |
| (iii)The cerebellum is much smaller in size as compared to the cerebrum. | The medulla is the connection between the brain and the spinal cord. |
| (iv) Planned actions like moving in a particular direction of swimming or riding a cycle are actions controlled by the cerebellum. | The blinking of the eyes is an involuntary action controlled by the medulla. |
Answer 19.
(a) The magnetic field produced around a straight current carrying conductor increases on increasing the strength of current in the conductor or vice-versa.
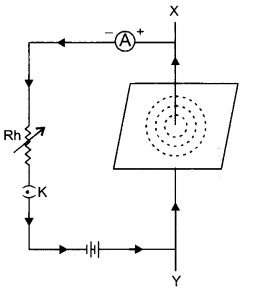
(b) Take a straight conductor XY and pass it through the centre of a cardboard. Connect the ends of the conductor to the terminals of a battery through a rheostat Rh, a key K and ammeter A, so that the current flows from Y to X, as shown in the figure given alongside:
Sprinkle some iron filings on the cardboard. When the cardboard is tapped, the iron filings will arrange themselves in concentric circles. These concentric circles will represent the magnetic field lines. This shows that around a straight current carrying conductor, there exist a magnetic field and the magnetic field is in the form of concentric circles with the conductor as centre.
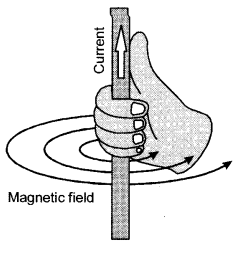
Maxwell’s right hand thumb rule is used to determine the direction of magnetic field for a known direction of current. If the thumb of the right hand points along the direction of current then the curled fingers of that hand gives the direction of the magnetic field due to the current. When thumb is upwards, the curled fingers are anticlockwise. So, magnetic field is anticlockwise. When thumb is downwards, the curled fingers are clockwise. So, magnetic field is clockwise.
(c) The two magnetic field lines can never intersect each other because two tangents can be drawn from that point of intersection which will give two directions of the field from the same point, which is impossible. So, the two magnetic field lines can never intersect each other.
Answer 20.
In Mendeleev’s periodic table, the elements are arranged in the order of their increasing atomic masses. In the Modem Periodic Table, the elements are arranged in the order of their increasing atomic numbers. Position of isotopes was not justified in Mendeleev’s periodic table while the Modem Periodic Table found a proper place for the isotopes. Position of hydrogen was a problem in r Mendeleev’s table while the Modem Periodic Table gave hydrogen a unique position due to its resemblance to both alkalies and halogens.
The trend in the Modem periodic Table with regard to valency is that elements are arranged in the order of increasing valency. Regarding atomic size it increases as we go down the group.
Answer 21.
A good source of energy should have the following features.
- It must do a large amount of work per unit volume or mass.
- It must be easily accessible.
- It must be easy to store and transport.
- It must be economical.
Some of the non-conventional sources of energy that man is trying to use these days are solar energy, tidal energy, wave energy, ocean thermal energy, geothermal energy and nuclear energy.
However these sources of energy need to be commercially viable and cost-effective in the long run. In India solar energy is a very viable option due to the availability of the bright sun for long hours during a major part of the year. However the cost of the solar cells is so high that at this point it is too expensive for domestic use. May be it could be a cost effective viable option in the future. Environ- [ mental consequence of using nuclear energy is finding a proper storage and disposal of spent or used fuels. Improper storage and disposal can lead to environmental contamination,
OR
(a) Fossil fuels are obtained by degradation of dead remains of plants and animals which are buried under the surface of earth over a million years ago. The are called so as they are produced by degradation of biomass over a million years ago.
(b) They are an important source of fuels. Petroleum products like petrol, diesel etc. are used as fuel in automobiles. Coal is used to thermal power plants to generate electricity and also used as a fuel for domestic purposes.
(c) The two fossil fuels are coal and petroleum.
(d) Methods of conserving fossil fuels are :
- Use of CNG in place of petrol and diesel as a fuel in automobiles.
- Switching to renewable sources of energy like solar energy, wind energy, hydroelectric energy instead of using non-renewable energy sources like fossil fuels.
- Wastage during extraction and transporation of fossil fuels should be minimized.
- Protecting them from accidental fires.
Over consumption and wastage of petrol or diesel should be checked in a automobiles by proper servicing and efficient internal combustion engines.
Answer 22.
When water is slowly added to calcium oxide or quick lime in a beaker the solution turns hot as heat is given off, because it is an exothermic reaction.
CaO(s) + H2O(I) → Ca(OH)2(aq)
Answer 23.
Tarnished copper vessels are washed with lemon or tamarind juice because the acid in the lemon or tamarind juice helps to remove the corrosion that has created a coating of brown or black colour on the copper vessel.
Answer 24.
The student saw the process of budding in yeast.
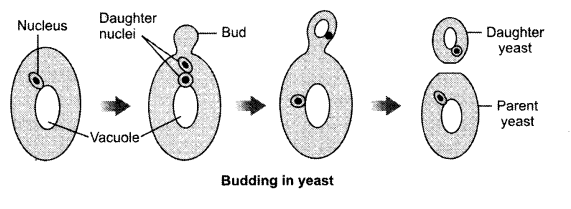
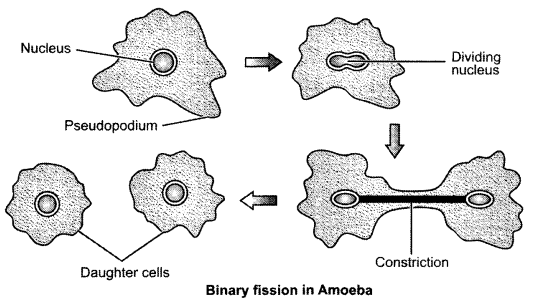
Answer 25.
Binary fission is the type of asexual reproduction in which two individuals are formed from a single parent and the parental identity is lost. This reproduction starts from division of nucleus i.e., karyokinesis.
Answer 26.
(a) Tina is suffering from myopia or short sightedness.
(b) Two possible causes of this defect are :
- Increase in the size of the eyeball.
- Decrease in the focal length of eye lens.
OR
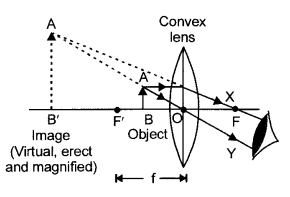
Answer 27.
If the student wants to connect four cells of 1.5 V each to form a battery of 6 V, then he has to connect them in series.

We hope the CBSE Sample Papers for Class 10 Science Paper 3 help you. If you have any query regarding CBSE Sample Papers for Class 10 Science Paper 3, drop a comment below and we will get back to you at the earliest.
