CBSE Sample Papers for Class 10 Science Paper 10
These Sample papers are part of CBSE Sample Papers for Class 10 Science. Here we have given CBSE Sample Papers for Class 10 Science Paper 10.
Time allowed : 3 hours
Maximum marks : 80
General Instructions
- The question paper comprises two sections, A and B. You are to attempt both the sections.
- All questions are compulsory.
- All questions of Section-A and B are to be attempted separately.
- There is an internal choice in two questions of three marks each and one question of five marks.
- Question numbers 1 and 2 in Section-A are one mark question. They are to be answered in one word or in one sentence.
- Question numbers 3 to 5 in Section-A are two marks questions. These are to be answered in 30 words each.
- Question numbers 6 to 15 in Section-A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section-A are 5 mark questions. These are to be answered in 70 words each.
- Question numbers 22 to 27 in Section-B are based on practical skills. Each question is a two marks question. These are to be answered in brief.
SECTION – A
Question. 1.
What category of diseases does AIDS come under ? Name its causative organism.
Question. 2.
Name the method by which Spirogyra reproduces under favourable conditions. Is this method sexual or asexual ?
Question. 3.
Dry hydrogen chloride gas does not turn blue litmus red whereas hydrochloric acid does. Give one reason.
Question. 4.
How can you distinguish between a plane mirror, a concave mirror and a convex mirror without touching them ?
Question. 5.
Where does the digestion of fat take place in our bodies ?
Question. 6.
A coil of insulated copper wire is connected to a galvanometer. What will happen if a bar magnet is
(a) pushed into the coil
(b) withdrawn from inside the coil
(c) held stationary inside the coil ?
OR
Two circular coils A and B are placed close to each other. If the current in the coil A is changed, will some current be induced in the coil B ? Give reason.
Question. 7.
Why is an ammeter likely to bum out if you connect it in parallel ?
Question. 8.
(a) What were the two major shortcomings of Mendeleev’s periodic table. How have these been removed in the modem periodic table ?
(b) Two elements X and Y have atomic numbers 12 and 16 respectively. Write the electronic configuration for these elements. To which period of the modem periodic table do these two elements belong ? What type of bond formed between them and why ?
Question. 9.
If the water supply at home requires a large amount of soap what do you infer ? Why does this happen ?
Question. 10.
What is biological magnification ? Are human beings affected by it ?
Question. 11.
What is the role of platelets in the human body ?
Question. 12.
An object placed on a meter scale at 8 cm mark was focussed on a white screen placed at 92 cm mark, using a converging lens placed on the scale at 50 cm mark.
(a) Find the focal length of the converging lens.
(b) Find the position of the image formed if the object is shifted towards the lens at a position of 29 cm.
(c) State the nature of the image formed if the object is further shifted towards the lens.
Question. 13.
Why do acids show acidic behaviour only in the presence of water ?
OR
A lowering of pH of the mouth is a major cause for tooth decay. Elaborate on this.
Question. 14.
Describe the female reproductive part of the flower.
Question. 15.
What is sustainable natural resource management ?
Question. 16.
Describe an activity to show that the colours of white light splitted by a glass prism can be recombined to get white light by another identical glass prism. Also draw ray diagram to show the recombination of the spectrum of white light.
Question. 17.
What are structural isomers ? Explain with examples.
OR
(a) What are hydrocarbons ? Give one example.
(b) Give the structural differences between saturated and unsaturated hydrocarbons with two examples each.|
(c) What is a functional group ? Give examples of two different functional groups.
Question. 18.
(a) Draw a neat labelled diagram showing the different endocrine glands in the human body.
(b) What happens when adrenaline is released into the blood ? Is it correct to call this hormone as the ‘fight or flight hormone’.
Question. 19.
Draw a labelled circuit diagram showing three resistors R1, R2 and R3 connected in series with a battery (E), a rheostat (Rh), a plug key (K) and an ammeter (A) using standard circuit symbol. Use this circuit to show that the same current flows through every part of the circuit. List two precautions you would observe while performing the experiment.
Question. 20.
How are ionic compounds formed ? Give their characteristics.
Question. 21.
What is the importance of forest as a resource ? What are the causes of deforestation ?
OR
Biomass is a good source of energy. Biogas is an excellent fuel. Elaborate.
SECTION – B
Question. 22.
If we pour acetic acid on red litmus paper and blue litmus paper, what happens ?
(a) Red litmus remains red and blue litmus turns red.
(b) Red litmus turns blue and blue litmus remains blue.
(c) Red litmus turns blue and blue litmus turns red.
(d) Red litmus turns colourless and blue litmus remains blue.
Question. 23.
While demonstrating decomposition reaction in the laboratory the teacher heated ferrous sulphate crystals in a hard glass dry boiling tube.
(a) What will be the smell of gases coming out of the boiling tube ?
(b) What will be the change in colour of the ferrous sulphate crystals ?
Question. 24.
Draw a labelled diagram of the cross-section of a leaf peel showing stomata.
Question. 25.
When you perform the experiment to show that CO2 is given out during respiration how will you confirm the gas evolved is CO2 ?
Question. 26.
The rest positions of the needles in a milliammeter and voltmeter not in use are as shown in Fig. A. When a student uses these in his experiment, the readings of the needle are in the positions shown in Fig. B.
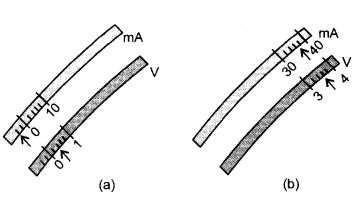
Find the corrected values of current and voltage in the experiment.
OR
The following circuit diagram shows the experimental set-up for the study of dependence of current on potential difference. Which two circuit components are connected in series ?
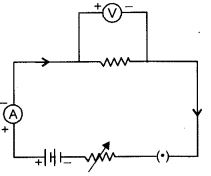
Question. 27.
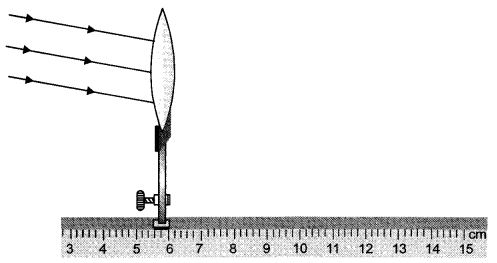
The teacher asks a student to fix the given screen at an appropriate place in the given experimental set up, so that a clear image can be obtained on the screen. If the focal length of convex lens in 8 cm, find the mark on the scale at which he should fix up the screen ?
ANSWERS
SECTION – A
Answer. 1.
AIDS comes under viral diseases which are sexually transmitted. It stands for Acquired Immuno Deficiency Syndrome and is caused by the Human Immuno Deficiency virus also called HIV virus.
Answer. 2.
Under favourable conditions, Spirogyra reproduces by fragmentation. This is an asexual method of reproduction.
Answer. 3.
Acids produce hydrogen ions [H+(aq)] in solution which are responsible for turning blue litmus red. Dry HCl does not give H+ ions in the absence of water.
Answer. 4.
By oberving the virtual images formed by the three mirrors, we can distinguish between the mirrors as :
- Plane mirror will produce an image of the same size,
- Concave mirror will produce a magnified image, and
- Convex mirror will produce a diminished image.
Answer. 5.
The small intestine is the site of the complete digestion of carbohydrates, proteins and fats. It receives the secretions of the liver and pancreas for this purpose. The food coming from the stomach is acidic and has to be made alkaline for the pancreatic enzymes to act. Bile juice from the liver accomplishes this in addition to acting on fats. Fats are present in the intestine in the form of large globules which makes it difficult for enzymes to act on them. Bile salts break them down into smaller globules to help enzymes act efficiently on them. The pancreatic juice contains enzymes like trypsin for digesting proteins and lipase for digesting emulsified fats. The enzymes in the intestinal juice secreted by the small intestine convert the proteins into amino acids, complex carbohydrates into glucose and fats into fatty acid and glycerol.
Answer. 6.
(a) When a bar magnet is pushed into the coil of insulated copper wire connected to a galvanometer, an induced current is set up in the coil. As a result, galvanometer gives a deflection (say towards left).
(b) When the bar magnet is withdrawn from inside the coil, again an induced current is set up in the coil. However, now direction of induced current is opposite to that in earlier case. Hence, deflection in galvanometer is in reverse direction (say towards right).
(c) If the bar magnet is held stationary inside the coil, then there is no induced current in the coil. As a result, galvanometer does not show any deflection.
OR
Yes, a current is induced in the coil B.
When the current in the coil A is changed, the magnetic field associated with it also changes. As coil B is placed close to A, hence magnetic field lines around this coil also change. Due to change in magnetic field lines associated with coil B, an induced current is also induced in it.
Answer. 7.
An ammeter has a very low resistance and is used to measure electric current flowing in a circuit. If an ammeter is connected in parallel with an electrical circuits, the net resistance of circuit becomes even less than the resistance of an ammeter and a strong current begins to flow. As a result the ammeter is likely to bum out.
(a) Two major shortcomings of Mendeleev’s periodic table :
- The position of isotopes could not be explained.
- Wrong order of atomic masses of some elements could not be explained.
In the modem periodic table, the elements are arranged in the increasing order of their atomic numbers instead of atomic masses.
(b) Atomic number of element X = 12
Electronic configuration = 2,8,2 KLM
Atomic number of element Y = 16
Electronic configuration = 2,8,6 KLM
The period number of an element is equal to the number of electron shells in its atom. These two elements have 3 electron shells, therefore they belong to 3rd period.
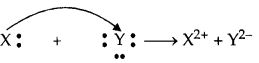
They will form ionic bond because X is a metal and Y is a non-metal. X loses two electrons which will be gained by Y.
Answer. 9.
If the water supply at home requires a large amount of water it means the water is hard i.e., it contains salts of calcium and magnesium. Usually the salts of calcium and magnesium cause hardness of water. Such type of water does not lather with soap. While bathing with hard water foam is formed with difficulty and an insoluble substance (scum) remains after washing with the water. This problem can be overcome by using another class of compounds called detergents as cleansing agents. Detergents are generally ammonium or sulphate salts of long chain carboxylic acids. The charged ends of these compounds do not form insoluble precipitates with the calcium and magnesium ions in hard water. Thus, they remain effective in hard water. Detergents are usually used to make shampoos and products for cleaning clothes.
Answer. 10.
Human beings are on top of any food chain. This means the maximum concentration of several harmful chemicals get accumulated in the human bodies. This is also called biological magnification. We have seen how water is polluted by different substances. Pesticides and some chemicals are used to protect crops from pests. These enter the soil and the water that flows on this soil. When this water is absorbed by the roots of plants it enters the plants. Similarly when aquatic animals drink water some chemicals enter their bodies too. These chemicals then enter the food chain. As they are not degraded they are gradually accumulated in each trophic level. Finally all of them reach the human bodies. Food grains like rice, wheat, vegetables, fruits, and meat contain varying amounts of pesticide residues. It is difficult to completely remove them by washing. Hence any organism at the top of the food chain is likely to get a fairly large amount of the harmful substances that enter the food chain at every level. So the effect of the various pollutants is maximised in human beings.
Answer. 11.
Platelets or thrombocytes are one of the constituents of blood the other two being the red blood,corpuscles or erythrocytes and white blood corpuscles or leucocytes. Whenever there is an injury in the body bleeding starts. The body cannot afford to lose blood. In addition, leakage of blood leads to a drop in pressure which affects the pumping of the heart. To avoid this, the body immediately informs the platelets to reach the site of injury. The platelets that keep circulating in the body quickly reach the site of bleeding and form a barrier or plug which stops the bleeding. That is why platelets,are needed by our body.
Answer. 12.
(a) Here position of object on meter scale = 8 cm, position of converging (convex) lens = 50 cm and position of screen = 92 cm.
u = (50 – 8) = 42 cm and υ = (92 – 50) = 42 cm
As per sign convention, u = – 42 cm and υ = + 42 cm.
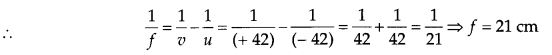
(b) If object is shifted to a position 29 cm, then u’ = (50 – 29) cm = 21 cm i.e., the object is situated at first focus Fa of the lens. Hence its real and inverted image is formed at infinity.
(c) If the object is further shifted towards the lens then | u | <f and hence a virtual and erect image of the obj ect will be formed.
Answer. 13.
Acids do not show acidic behaviour in the absence of water. This is because the H+ ions are produced only in the presence of water. The H+ ions cannot exist alone but they exist only after combining with water molecules.
If water is added to HCI the reaction occurs as follows :
HCI + H2O → H3O+ + Cl–
The separation of H+ ions from the HCI molecule cannot occur in the absence of water. The process of dissolving acid in water is highly exothermic. The acid must be slowly added to the water with constant stirring, water must not be added to the acid.
OR
A lowering of the pH of the mouth means the environment inside the mouth is turning acidic. We have learnt that when the pH is less than 7 the said material or solution is acidic. When acids are formed in the mouth due to the deposit of leftover food particles between the teeth it leads to a slow destruction of the enamel of the teeth. Initially it appears as a black spot but soon it grows in size and destroys the entire enamel. Once the enamel is removed the nerves and blood vessels under the teeth are exposed. This is when we experience severe pain whenever anything hot or cold is ingested.
Answer. 14.
The carpel is the female reproductive part of the flower which is present in the centre of the flower. It consists of three parts. The upper part which is sticky and is called the stigma, the middle elongated part is called the style and the swollen bottom part is called ovary. The ovary contains the ovules and each ovule has an egg inside. The male germ-cell produced by the pollen grain fuses with the female germ cell in the ovule. This is called fertilisation and it results in the formation of the zygote. This zygote grows into a new plant.
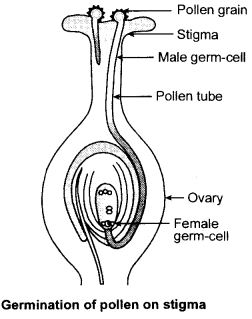
Answer. 15.
All the things we use or consume like food, clothes, our shelter, books, toys, furniture, tools and vehicles are obtained from resources on this earth. The only thing we get from outside is the energy from the sun. This too is processed by the living organisms and various physical and chemical processes on the earth before we can use it. We should use resources judiciously because they are limited. We have an ever-increasing human population due to improved health care and this has brought about an increased demand for all the resources. The management of natural resources requires a long-time perspective so that they will last for the future generations and will not be exploited over a short period of time. Resource management must be focussed on an equitable distribution of resources so that it is not just a handful of rich and powerful people who benefit from this. In addition, when we extract these resources we must ensure a safe disposal of the wastes too. This is called sustainable natural resource management.
Answer. 16.
Recombination of colours: The colours of white light splitted by a glass prism can be recombined to get white light by another identical glass prism. Newton demonstrated this phenomenon of recombination of the coloured rays of a spectrum to get back white light.
- A triangular prism ABC is placed on its base BC.
- A similar prism A’B’C’ is placed alongside with its refracting surface in the opposite direction, i.e., in an inverted position with respect to first prism as shown in figure.
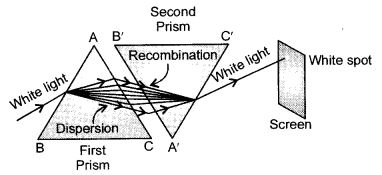
- A beam of white light entering the prism ABC undergoes refraction and is dispersed into its constituent seven colours.
- These constituent seven coloured rays are incident on the second inverted prism A’B’C’ and get further refracted.
- The second prism recombines them into a beam of white light and emerges from the other side of the second prism and falls on the screen.
- This is due to the fact that the refraction or bending produced by the second inverted prism is equal and opposite to the refraction or bending produced by the first prism. This causes the seven colours to recombine.
- A white patch of light is formed on the screen placed beyond the second prism. This proves the phenomenon of recombination of spectrum of white light.
Answer. 17.
Structural isomers are carbon compounds that have the same or identical molecular formula but different structural formula. An example of structural isomers is shown below :
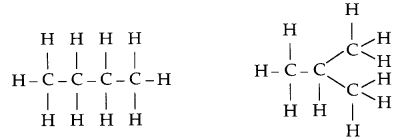
In addition to straight and branched carbon chains, some compounds have carbon atoms arranged in the form of a ring. Carbon has the unique ability to form bonds with other atoms of carbon giving rise to large molecules. This property is called catenation. We can find compounds with long chains of carbon or even carbon atoms arranged in a ring. In addition carbon atoms can be linked by single, double or triple bonds. As carbon is tetravalent it forms covalent bonds with four other atoms of carbon or atoms of some other monovalent element. These bonds are very strong and so the elements are very stable. Compounds of carbon which are linked by only single bonds between carbon atom are called saturated compounds. Compounds of carbon having double or triple bonds between their carbon atoms are called unsaturated compounds.
OR
(a) Compounds of carbon and hydrogen are called hydrocarbons, e.g., methane, ethane, etc.
(b) Saturated hydrocarbons contains carbon-carbon single bonds, e.g., methane, ethane. Unsaturated hydrocarbons contain atleast one carbon-carbon double or triple bond, e.g., ethene, ethyne.
(c) Functional group is an atom or group of atoms joined in a specific manner which is responsible for the characteristics properties of the organic compounds, e.g., OH, – CHO.
Answer. 18.
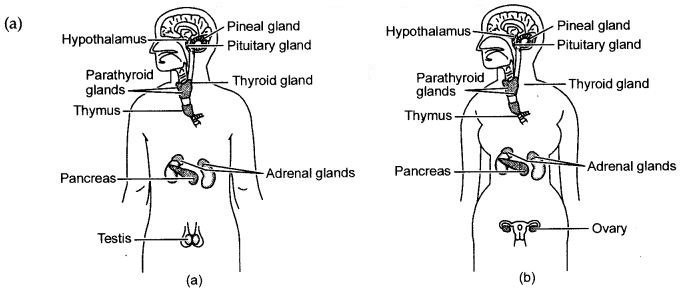
(b) Hormones are chemical substances secreted by ductless or endocrine glands in the body. One such hormone is adrenaline. It is secreted by the adrenal glands which areb present just above the kidneys. Adrenaline is directly released into the blood and carried to different parts of the body. The target organs on which it acts includes the heart. As a result, the heart beats faster, resulting in supply of more oxygen to our muscles. The blood to the digestive system and skin is reduced due to contraction of muscles around small arteries in these organs. This diverts the blood to the skeletal muscles. There is an increase in the breathing rate too. All these responses make the animal body ready to face the situation. The animal either faces the situation or flees from danger. Hence adrenaline is called the fight or flight hormone.
Answer. 19.
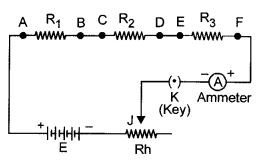
Aim : Same current flows through every part of the above circuit.
Procedure :
- Connect the another ammeters, ‘A1‘ between B and C, and ‘A2‘ between D and E.
- Adjust the sliding contact ‘j’ of the rheostat initially for a small current.
- Note all the ammeter readings. This reading gives us current flowing through the resistors R1,R2 and R3.
- The current in the circuit is now increased by changing the position of sliding contact ‘j’ of the rheostat.
- Note all the ammeter readings each time.
Conclusion : Same reading of all the ammeter in each observation concluded that same current flows through every part of the circuit.
Precautions:
- All the connection should be tight and properly connected as per circuit diagram.
- The positive terminal of the ammeter and voltmeter must be connected to the positive terminal of the battery or battery eliminator.
Answer. 20.
Ionic compounds are formed by the transfer of electrons from one atom to another in an effort to get the stable electronic configuration in their outermost shell. The bond formed is called electrovalent bond and the atoms in that are either positive or negative ions. These ions are held together by strong forces of attraction. Hence ionic compounds are usually solids.
- Ionic compounds have high melting and boiling points this means a lot of energy is required to break the strong inter-ionic attraction.
- A solution of ionic compounds contains ions which move to the oppositely charged electrodes when current passes through the solution. Ionic compounds do not conduct electricity in the solid state as ions cannot move about. Ionic compounds conduct electricity in the molten state as the electrostatic forces between the ions is overcome by the heat.
Answer. 21.
Forests are an important natural resource due to following reasons :
- Forests provide us wood, dry fruits, spices, gums, resins, dyes, tannins and various products like honey, camphour, rubber etc.
- The roots of trees hold the soil firmly preventing soil erosion.
- It absorbs carbon dioxide from atmosphere and release oxygen which is life supporter.
- It absorbs carbon dioxide which is a greenhousfe gas thus plays an important role in reducing global warming.
- It increases frequency of rainfall and regulates water cycle.
- It serves as a shelter for wildlife.
Causes of deforestation are :
- Large scale deforestation for demand of wood, timber as raw materials for industries, domestic purposes etc.
- Construction of dams, reservoirs, roads, railways.
- Setting up of factories and industries.
- Construction of big apartments to provide space to the growing population.
- Shifting cultivation for agriculture.
- Overgrazing by livestock.
OR
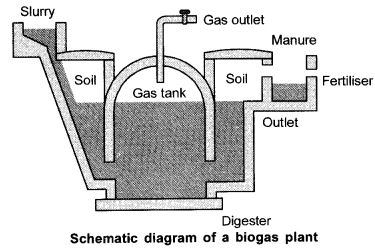
While looking at the conventional sources of energy a number of points can be considered. Wood has been used as a fuel for a long time. This can continue if only we have enough trees around to provide a continuous supply of wood. Even cow-dung cakes are used as fuel. With a large livestock population in our country cow-dung cake can also be used as a steady source of fuel. As both these fuels are from plant and animal products, the source of these fuels is said to be biomass. These fuels, however, do not produce much heat on burning and a lot of smoke is given out when they are burnt. So, one has to look out for new technological inputs to improve the efficiency of these fuels. When wood is burnt in a limited supply of oxygen, water and other volatile substances in it are removed and only charcoal is left behind as residue. Charcoal bums without flames, is comparatively smokeless and has higher heat generation efficiency.
Cow-dung, plant residue after harvesting of crops, vegetable waste and sewage are decomposed in the absence of oxygen to give bio-gas. As the starting material is mainly cow-dung, it is popularly known as gobar-gas. Biogas is produced in the biogas plant biogas is an excellent fuel as it contains upto 75% methane. It bums without smoke, leaves no residue like ash in wood, charcoal and coal burning. Its heating capacity is high. Biogas is also used for lighting. The slurry left behind is removed periodically and is used as excellent manure, rich in nitrogen and phosphorous. The large-scale utilization of bio¬waste and sewage material provides a safe and efficient method of waste-disposal besides supplying energy and manure.
SECTION – B
Answer. 22.
(a) Red litmus remains red and blue litmus turns red.
Answer. 23.
(a) There will be no smell of gases coming out of the boiling tube.
(b) The light green crystals of ferrous sulphate will turn white in colour.
Answer. 24.
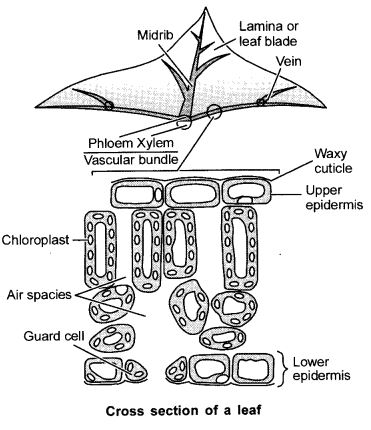
Answer. 25.
The gas that is collected at the end of the process is allowed to pass through lime water. When lime water turns milky it is a confirmation that the gas is CO2.
Answer. 26.
Milliammeter has negative zero error and voltmeter has positive zero error.
Zero correction is always, negative of zero error.
∴ Milliammeter reading = 38 – (- 4) = 38 + 4 = 42 mA
Voltmeter reading = 3.6 – (+ 0.4)
= 3.6 – 0.4 = 3.2 V
OR
Ammeter and rheostat are connected in series.
This is because only one common point exists between ammeter, cell and rheostat.
Answer. 27.
When the object is at infinity from a convex lens, then the image formed is at focus (on the other side of the lens). The lens with stand is fixed at 5.8 cm. The focal length of convex lens is 8 cm. Therefore, he should fix up the screen at 13.8 cm mark on the scale.
We hope the CBSE Sample Papers for Class 10 Science paper 10 help you. If you have any query regarding CBSE Sample Papers for Class 10 Science paper 10, drop a comment below and we will get back to you at the earliest.
