These NCERT Solutions for Class 9 Science Chapter 1 Matter in Our Surroundings Questions and Answers are prepared by our highly skilled subject experts to help students while preparing for their exams.
Matter in Our Surroundings NCERT Solutions for Class 9 Science Chapter 1
Class 9 Science Chapter 1 Matter in Our Surroundings InText Questions and Answers
Question 1.
Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold, cold drink, smell of perfume.
Answer:
Anything that occupies space and has mass is called matter. Matter can exist in three physical states—solid, liquid, and gaseous.
Chair and almond are forms of matter in the solid state.
Cold drink is a liquid state of matter.
Air is a gaseous state of matter.
Note: The sense of smell is not matter. However, the smell or odour of a substance is classified as matter. The smell of any substance (say, perfume) can be classified as matter. This is because, perfume is in liquid state inside the bottle.
It comes out in the form of tiny droplets. These droplets of perfume are matter and spread out in the atmosphere to create a sense of smell. This smell can be detected by our olfactory system.
Question 2.
Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Answer:
Solids diffuse at a very slow rate. But, if the temperature of the solid is increased, then the rate of diffusion of the solid particles into air increases. This is due to an increase in the kinetic energy of solid particles. Hence, the smell of hot sizzling food reaches us even at a distance, but to get the smell from cold food we have to go close.
Question 3.
A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer:
The ability of a diver to cut through water in a swimming pool shows that the particles of matter have intermolecular spaces. The intermolecular spaces in liquids is fair enough to let the diver pass through it.
Question 4.
What are the characteristics of particles of matter?
Answer:
The characteristics of particles of mat¬ter are:
- Particles of matter have spaces between them.
- Particles of matter are continuously moving.
- Particles of matter attract each other.
Question 5.
The mass per unit volume of a substance is called density (density = mass/volume). Arrange the following in order of increasing density – air, exhaust from chimney, honey, water, chalk, cotton, and iron.
Answer:
The given substances in the increasing order of their densities can be represented as:
Air < Exhaust from chimney < Cotton < Water < Honey < Chalk < Iron
![]()
Question 6.
(a) Tabulate the differences in the characteristics of states of matter.
(b) Comment upon the following: rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy, and density.
Ans. (a) The differences in the characteristic states of matter are given in the following table.
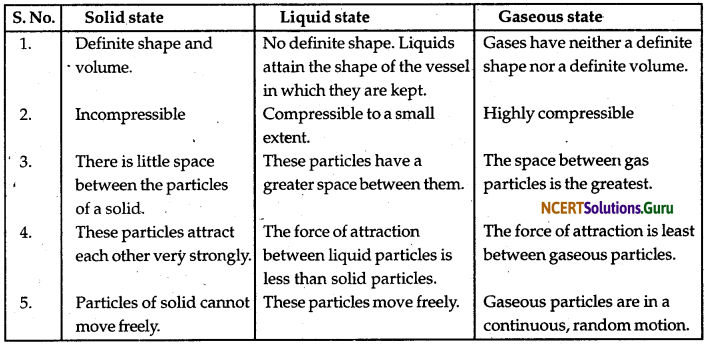
(b) Rigidity can be expressed as the tendency of matter to resist a change in shape. Compressibility is the ability to be reduced to a lower volume when force is applied.
- Fluidity is the ability to flow.
- By filling a gas container we mean the attainment of shape of the container by gas.
- Shape defines a definite boundary.
- Kinetic energy is the energy possessed by a particle due to its motion.
- Density is mass per unit volume.
Question 7.
Give reasons:
(a) A gas fills completely the vessel in which it is kept.
(b) A gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air, but to do the same through a solid block of wood, we need a karate expert.
Answer:
(a) There is little attraction between particles of gas. Thus, gas particles move freely in all directions. Therefore, gas completely fills the vessel in which it is kept.
(b) Particles of gas move randomly in all directions at high speed. As a result, the particles hit each other and also hit the walls of the container with a force. Therefore, gas exerts pressure on the walls of the container.
(c) A wooden table has a definite shape and volume. It is very rigid and cannot be compressed i.e., it has the characteristics of a solid. Hence, a wooden table should be called a solid.
(d) Particles of air have large spaces between them. On the other hand, wood has little space between its particles. Also, it is rigid. For this reason, we can easily move our hands in air, but to do the same through a solid block of wood, we need a karate expert.
Question 8.
Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
Answer:
The mass per unit volume of a substance is called density (density = mass/volume). As the volume of a substance increases, its density decreases.
Though ice is a solid, it has large number of empty spaces between its particles. These spaces are larger as compared to the spaces present between the particles of water. Thus, the volume of ice is greater than that of water. Hence, the density of ice is less than that of water. A substance with lower density than water can float on water. Therefore, ice floats on water.
![]()
Question 9.
Convert the following temperature to Celsius scale:
(a) 300 K (b) 573 K
Answer:
(a) 300 K = (300 – 273)°C = 27°C
(b) 573 K = (573 – 273)°C = 300°C
Question 10.
What is the physical state of water at:
(a) 250°C
(b) 100°C
Answer:
(a) Water at 250°C exists in gaseous state.
(b) At 100°C, water can exist in both liquid and gaseous form. At this temperature, after getting the heat equal to the latent heat of vaporization, water starts changing from liquid state to gaseous state.
Question 11.
For any substance, why does the temperature remain constant during the change of state?
Answer:
During a change of state, the temperature remains constant. This is because all the heat supplied to increase the temperature is utilised in changing the state by overcoming the forces of attraction between the particles. Therefore, this heat does not contribute in increasing the temperature of the substance.
Question 12.
Suggest a method to liquefy atmospheric gases.
Answer:
By applying pressure and reducing the temperature, atmospheric gases can be liquefied.
Question 13.
Why does a desert cooler cool better on a hot dry day?
Answer:
When a liquid evaporates, the particles of the liquid absorb energy from the surroundings to compensate the loss of energy during evaporation. This makes the surroundings cool.
In a desert cooler, the water inside it is made to evaporate. This leads to absorption of energy from the surroundings, thereby cooling the sur¬roundings. Again, we know that evaporation depends on the amount of water vapour present in air (humidity). If the amount of water vapour present in air is less, then evaporation is more. On a hot dry day, the amount of water vapour present in air is less. Thus, water present inside the desert cooler evaporates more, thereby cooling the surroundings more. That is why a desert cooler cools better on a hot dry day.
Question 14.
How does water kept in an earthen pot (matka) become cool during summers?
Answer:
There are some pores in an earthen pot through which the liquid inside the pot evaporates. This evaporation makes the water inside the pot cool. In this way, water kept in an earthen pot becomes cool during summers.
Question 15.
Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Answer:
When we put some acetone or petrol or perfume on our palm, it evaporates. During evaporation, particles of die liquid absorb energy from the surrounding or the surface of the palm to compensate for the loss of energy, making the surroundings cool. Hence, our palm feels cold when we put some acetone or petrol or perfume on it.
![]()
Question 16.
Why are we able to sip hot tea or milk faster from a saucer than a cup?
Answer:
A liquid has a larger surface area in a saucer than in a cup. Thus, it evaporates faster and cools faster in a saucer than in a cup. For this reason, we are able to sip hot tea or milk faster from a saucer than a cup.
Question 17.
What type of clothes should we wear in summers?
Answer:
We should wear cotton clothes in summers. During summers, we sweat more. On the other hand, cotton is a good absorber of water. Thus, it absorbs sweat from our body and exposes the liquid to the atmosphere, making evaporation faster. During this evaporation, particles on the surface of the liquid gain energy from our body surface, making the body cool.
Class 9 Science Chapter 1 Matter in Our Surroundings Textbook Questions and Answers
Question 1.
Convert the following temperatures to Celsius scale.
(a) 293 K
(b) 470 K
Answer:
Kelvin is an SI unit of temperature, where 0°C = 273.16 K (approximately 273 K)
(a) 293 K = (293 – 273) °C = 20 °C
(b) 470 K = (470 – 273) °C= 197 °C
Question 2.
Convert the following temperatures to Kelvin scale.
(a) 25°C
(b) 373°C
Answer:
Kelvin is an SI unit of temperature, where 0°C = 273.16 K (approximately 273 K)
(a) 25 °C = (25 + 273) K = 298 K
(b) 373 °C = (373 + 273) K = 646 K
Question 3.
Give reason for the following observations.
(a) Naphthalene balls disappear with time without leaving any solid.
(b) We can get the smell of perfume sitting several metres away.
Answer:
(a) Naphthalene undergoes sublimation easily i.e., the change of state of naphthalene from solid to gas takes place easily. Thus, naphthalene balls disappear with time without leaving any solid.
(b) Gaseous particles possess high speed and large spaces between them. Particles of perfume diffuse into these gaseous particles at a very fast rate and reach our nostrils. This enables us to smell the perfume from a distance.
![]()
Question 4.
Arrange the following substances in increasing order of forces of attraction between particles-water, sugar, oxygen.
Answer:
Sugar is a solid; the forces of attraction between the particles of sugar are strong. Water is a liquid; the forces of attraction here are weaker than sugar. Oxygen is a gas; the forces of attraction are the weakest in gases.
Thus, the increasing order of forces of attraction between the particles of water, sugar and oxygen is
Oxygen < Water < Sugar
Question 5.
What is the physical state of water at
(a) 25°C
(b) 0°C
(c) 100°C
Answer:
(a) Water at 25°C is present in the liquid state.
(b) At 0°C, water can exist as both solid and liquid. At this temperature, after getting the heat equal to the latent heat of fusion, the solid form of water i.e., ice starts changing into its liquid form i. e., water.
(c) At 100 °C, water can exist as both liquid and gas. At this temperature, after getting the heat equal to the latent heat of vaporization, water starts changing from its liquid state to its gaseous state, i.e., water vapours.
Question 6.
Give two reasons to justify that
(a) water at room temperature is a liquid.
(b) an iron almirah is a solid at room temperature.
Answer:
(a) At room temperature (25 °C), water is a liquid because it has the following characteristic of liquid:
- At room temperature, water has no shape but has a fixed volume that is, it occupies the shape of the container in which it is kept.
- At room temperature, water flows.
(b) An iron almirah is a solid at room temperature (25 °C) because:
- it has a definite shape and volume like a solid at room temperature.
- it is rigid as solid at room temperature.
Question 7.
Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer:
Ice at 273 K has less energy than water (although both are at the same temperature). Water possesses the additional latent heat of fusion. Hence, at 273 K, ice is more effective in cooling than water.
Question 8.
What produces more severe bums, boiling water or steam?
Answer:
Steam has more energy than boiling water. It possesses the additional latent heat of vaporization. Therefore, bums produced by steam are more severe than those produced by boiling water.
Question 9.
Name A, B, C, D, E and F in the following diagram showing change in its state.
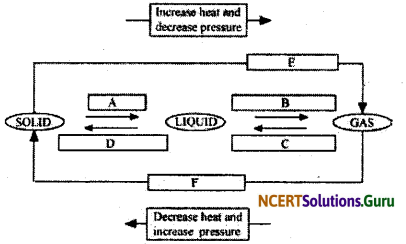
Answer:
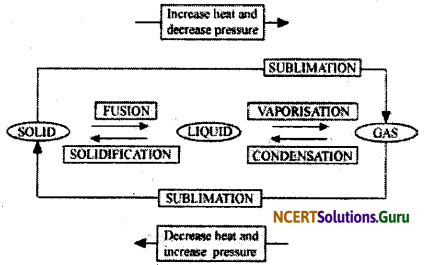
![]()
Class 9 Science Chapter 1 Matter in Our Surroundings Additional Important Questions and Answers
Multiple Choice Questions
Choose the correct option:
Question 1.
Which one of the following sets of phenomena would increase on raising the temperature?
(a) Diffusion, evaporation, compression of gases
(b) Evaporation, compression of gases, solubility
(c) Evaporation, diffusion, expansion of gases
(d) Evaporation, solubility, diffusion, compression of gases
Answer:
(c) Evaporation, diffusion, expansion of gases
Question 2.
Seema visited a Natural Gas Compressing Unit and found that the gas can be liquefied under specific conditions of temperature and pressure. While sharing her experience with friends she got confused. Help her to identify the correct set of conditions
(a) Low temperature, low pressure
(b) High temperature, low pressure
(c) Low temperature, high pressure
(d) High temperature, high pressure
Answer:
(c) Low temperature, high pressure
Question 3.
The property to flow is unique to fluids. Which one of the following statements is correct?
(a) Only gases behave like fluids
(b) Gases and solids behave like fluids
(c) Gases and liquids behave like fluids
(d) Only liquids are fluids
Answer:
(c) Gases and liquids behave like fluids
Question 4.
During summer, water kept in an earthen pot becomes cool because of the phenomenon of
(a) diffusion
(b) transpiration
(c) osmosis
(d) evaporation
Answer:
(d) evaporation
![]()
Question 5.
A few substances are arranged in the increasing order of forces of attraction between their particles. Which one of the following represents a correct arrangement?
(a) Water, air, wind
(b) Air, sugar, oil.
(c) Oxygen, water, sugar
(d) Salt, juice, air
Answer:
(c) Oxygen, water, sugar
Question 6.
On converting 25°C, 38°C and 66°C to kelvin scale, the correct sequence of temperature will be
(a) 298 K, 311 K and 339 K
(b) 298 K, 300 K and 338 K
(c) 273 K, 278 K and 543 K
(d) 298 K, 310 K and 338 K
Answer:
(a) 298 K, 311 K and 339 K
Question 7.
Choose the correct statement of the following
(a) conversion of solid into vapours without passing through the liquid state is called vapourisation.
(b) conversion of vapours into solid without passing through the liquid state is called sublimation.
(c) conversion of vapours into solid without passing through the liquid state is called freezing.
(d) conversion of solid into liquid is called sublimation.
Answer:
(b) conversion of vapours into solid without passing through the liquid state is called sublimation.
Question 8.
The boiling points of diethyl ether, acetone and n-butyl alcohol are 35°C, 56°C and 118°C respectively. Which one of the following correctly represents their boiling points in kelvin scale?
(a) 306 K, 329 K, 391K
(b) 308 K, 329 K, 392 K
(c) 308 K, 329 K, 391 K
(d) 329 K, 392 K, 308 K
Answer:
(c) 308 K, 329 K, 391 K
Question 9.
Which condition out of the following will increase the evaporation of water?
(a) Increase in temperature of water
(b) Decrease in temperature of water
(c) Less exposed surface area of water
(d) Adding common salt to water
Answer:
(a) Increase in temperature of water
Question 10.
In which of the following conditions, the distance between the molecules of hydrogen gas would increase?
(i) Increasing pressure on hydrogen contained in a closed container
(ii) Some hydrogen gas leaking out of the container
(iii) Increasing the volume of the container of hydrogen gas
(iv) Adding more hydrogen gas to the container without increasing the volume of the container
(a) (i) and (iii)
(b) (i) and (iv)
(c) (ii) and (iii)
(d) (ii) and (iv)
Answer:
(c) (ii) and (iii)
![]()
Very Short Answer Questions
Question 1.
Name any two things that are not the state of matter.
Answer:
The feelings of love and hate are not the state of matter.
Question 2.
Name any two substances which exist in all the three states of matter at low range of temperature.
Answer:
Water and wax.
Question 3.
Define the specific heat of a substance.
Answer:
It is the amount of heat required to raise the temperature of a gram of substance by 1°C.
Question 4.
How is the boiling point of a liquid affected with the increasing or decreasing pressure?
Answer:
The boiling of a liquid is proportional to the pressure i.e. with the increasing pressure, boiling point will increase and with the decreasing pressure, it will decrease.
Question 5.
What is the advantage of boiling the acids under low pressure conditions?
Answer:
It helps to concentrate them without them undergoing any decomposition.
Question 6.
What is the S.I. unit for expressing the specific heat of a substance?
Answer:
Cal/g/°C
![]()
Question 7.
The calorie and joule are the units used to measure the heat energy. How are they interrelated?
Answer:
1 Joule = 1 / 4.18 calorie or 1 calorie = 4.18 Joules
Question 8.
What happens when a body warms up?
Answer:
When a body warms up showing the rise in temperature, it absorbs the heat from its surroundings.
Question 9.
What happens when a hot body is left at room temperature to cool?
Answer:
When a hot body is left at room temperature to cool, it looses the heat to the surroundings.
Question 10.
What effect does the evaporation of a liquid has on the liquid?
Answer:
Evaporation of a liquid has cooling effect i.e., the temperature of a liquid falls with increasing evaporation.
Question 11.
Why are the earthen pots more effective in Cooling water in rural areas?
Answer:
In rural areas where there is no electricity, the earthen pots are effective means for cooling water because they allow the evaporation of water from the surface.
Question 12.
When will the clothes spread on a cloth line will dry faster, on a hot humid day or hot dry day?
Answer:
On hot dry day as the relative atmospheric humidity is less, the rate of evaporation will be more and hence the domes will dry faster.
Question 13.
Alcohol and water are two liquids. Alcohol has lower boiling point than water. Which of the two would evaporate faster at a given temperature?
Answer:
Alcohol, it has low boiling point as compared to the water.
Question 14.
State the two precautions you will observe when measuring the volume of a liquid using a measuring cylinder?
Answer:
The two precautions include placing the measuring cylinder on the flat surface and reading the lower meniscus if water, with eyes in line of the meniscus.
Question 15.
What is dry ice?
Answer:
It is the solidified form of carbon dioxide at low temperature and high pressure conditions.
![]()
Question 16.
What are the three properties of the state of matter?
Answer:
Mass, volume and inertia are the three properties of states of matter.
Question 17.
What happens to the particles of a matter when it is heated?
Answer:
The particles of matter gain the thermal energy and use it to increase their kinetic energy.
Question 18.
Why do we feel our palm cool after placing an ice cube on it?
Answer:
Ice cube placed on palm extracts the heat from the palm surface to melt. This extraction of heat cools the palm.
Question 19.
What is a pure substance?
Answer:
A substance which has its specific melting point, boiling point and density is called a pure substance. All the elements and compounds are examples of pure substance.
Question 20.
What are volatile liquids?
Answer:
Liquids which vapourise even at the room temperature are called volatile liquids for example alcohol, acetone, etc.
Short Answer Questions
Question 1.
What is matter? State its different forms.
Answer:
Anything in our surroundings that occupies space and has its own mass is called matter. All forms of the matter are made of the small particles that are invisible to naked eyes.
Question 2.
What happens when 10g of table salt is dissolved in the 100 ml of water? Does it bring any change in the volume of the solution?
Answer:
When salt is added to water, the particles of the salt occupies the spaces present in between the particles of the water hence, their occur no evident change in its volume.
Question 3.
A student mixed 100 ml alcohol and 100 ml of water together in a container. What would be the net volume of mixture in the container? Why?
Answer:
The net volume of the mixture in the container would be slightly less than 200 ml because the particles of the water and alcohol would occupy the spaces present between each other.
Question 4.
How will you experimentally prove that matter is made up of small particles?
Answer:
Take a known volume of water in a container and dissolve a small amount of table salt in water by continuous stirring. When the volume of the solution is measured, it shows no difference to show that the matter is made of small particles with spaces among them that can be occupied by the others.
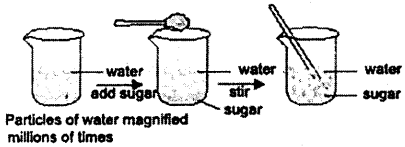
![]()
Question 5.
(a) What is diffusion?
(b) State the different factors which affect the diffusion.
Answer:
(a) Diffusion refers to the phenomenon of the intermixing of particles of the two or more substances on their own based on the concentration differences.
(b) Diffusion is affected by the concentration differences, mass of the diffusing particles, the medium in which the particles have to diffuse and the distance in between the source and sink.
Question 6.
Why do you smell a lighted incense stick put away in the house temple on ground floor when studying in your study room on the first floor?
Answer:
The particles of perfume in incense stick mix with the air and freely move in all directions from the area of their higher concentration in the temple room to the other rooms. The incoming of these perfume particles because of their diffusion, makes you smell the sweet smell of incense.
Question 7.
What are the different states of matter? Give examples.
Answer:
There are three states of matter, solid, liquid and gases.
- Solid: Stone, brick, salt, sugar, a metal block, etc.
- Liquid: liquid water, alcohol, milk, oil, petrol, etc.
- Gases: Oxygen, nitrogen, hydrogen, carbon dioxide, etc.
Question 8.
Classify the constituents of body into the different states of matter.
Answer:
Solid: Bones, teeth and muscles.
Liquid: Water, blood and lymph Gases: Inhaled air and exhaled air.
Question 9.
A rubber band can be stretched unlike solids then why shall it be considered a solid?
Answer:
A rubber band is a solid despite of the fact that it can be stretched because it does not change its shape at its own. An external force needs to be applied on it to force it change its shape and after the removal of the force, it mostly regains its shape.
Question 10.
A handful of salt or sugar take the shape of the container like liquids then why are they considered solids than liquids?
Answer:
A handful of salt or sugar does take the shape of the container like liquids yet they are considered solids because the individual crystal they contain, has a fixed shape.
![]()
Question 11.
Why is a sponge considered a solid when it can be compressed like gases or liquids?
Answer:
A sponge is considered solid even when it can be compressed like gases or liquids because its compression is because of the presence of minute air holes or cavities filled with air rather than the solid matter it has in its structure.
Question 12.
Define volume? Write its S.I. unit.
Answer:
Volume is the space occupied by a substance. All the solids and liquids have definite volume. It’s S.I. unit cubic metre (m3) but commonly used in cm3 when, 1 cm3 = 1000 ml = 1 litre.
Question 13.
Define density. State its S.I. unit.
Answer:
Density refers to the mass per unit
volume of a substance. It is measured in kg/m3 or g/cm3.
Density = \(\frac{\text { Mass }}{\text { Volume }}\)
Question 14.
Which have more density, solids or liquids? Why?
Answer:
Solids have more density than liquids because with particles placed close together, they have more mass per unit volume as compared to liquids in which the particles are loosely packed.
Question 15.
Why gases can be compressed but not solids?
Answer:
The gases can be compressed because their particles have very less force of attraction in between them so that they are in random motion with large distance among one another. Hence, when external pressure is applied, the particles come close to each other and the gas compresses but in solids the particles have large force of attraction hence, the distance is very less such that even upon applying the external pressure, they fail to be compressed.
Question 16.
Can particles of a solid diffuse into another solid? If yes, give example.
Answer:
Yes, the particles of a solid can diffuse into another solid but the diffusion takes a long time. For example if two metal blocks are tied together and left undisturbed for a long time i.e. for years, the particles of one diffuse into another as it become difficult to separate mem from each other.
![]()
Question 17.
What do you understand by the compression of a gas? Give examples of the use of compressed gas in daily life.
Answer:
The compression of a gas refers to reducing of the volume of a gas in a container on applying external pressure. The different compressed gases are used in daily life such as LPG (Liquefied petroleum gas) used for cooking, oxygen gas used in hospitals and CNG (Compressed natural gas) used as fuel in vehicles.
Question 18.
Why liquids have definite volume but not shape?
Answer:
Liquids have definite volume but not shape because they have moderate force of attraction between particles such that the distance between the particles is also moderate giving them a definite volume but not shape.
Question 19.
What do you understand by the anomalous behaviour of water? What is its advantage to living organisms?
Answer:
Water has anomalous behaviour because unlike other substances, on freezing its density decreases and therefore the ice floats on the water surface. The floating of ice is important for aquatic living organisms as they can survive in water below the surface of thick ice in oceans such as Arctic ocean and polar regions.
Question 20.
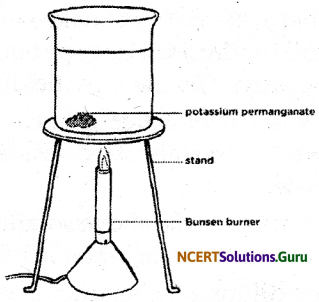
What happens when a crystal of potassium permanganate is gently placed at the bottom of the water rilled beaker?
Answer:
The crystal of potassium permanganate gets dissolved in water. The dissolution involves the diffusion of its particles in water. This diffusion continues until the particles of potassium permanganate get evenly distributed in water.
Question 21.
State the relation in between the density and diffusion of a given substance into another substance.
Answer:
Diffusion involves the free intermixing of a substance into another. It is inversely proportional to the under root of the density of the substance. Therefore, an ink with low density dissolves or diffuses faster in water than a drop of honey which is denser than ink.
Question 22.
How will you test the purity of honey bought from a shop?
Answer:
A drop of the honey to be tested is poured in a glass of water. If it forms a streak going down the water than its pure, otherwise not.
Question 23.
How do the aquatic plants and animals manage to survive in water?
Answer:
All aquatic plants and animals require carbon dioxide or oxygen or both for their successful survival. These gases are soluble in water. Carbon dioxide dissolves in water forming carbonic acid which is a very acid weak and oxygen is sparingly soluble in water, about 0.7% per litre of water.
Question 24.
Arrange the following
Liquid, solid, gas
in the increasing order of the rate of diffusion of their particles with reason.
Answer:
In solids the force of attraction are very strong hence, the particles are” bound close to each other but in gases, the force of attraction between the particles is very less, hence, they are free to move randomly. Therefore, in gases the rate of the diffusion is maximum than in solids.
The liquids have moderate force of attraction, therefore they lie in between solids and liquids for the rate of diffusion.
Solids < Liquids < Gases
![]()
Question 25.
Why does a gas stored in the container exerts pressure on its walls?
Answer:
A gas stored in a container exerts pressure on its walls because the gas particles are in free random motion. Whenever, they hit the wall of the container, they exert pressure which is proportional to the motion of the particles and their density.
Question 26.
Why does an inflated balloon when kept in sun expands in size and when kept in deep freezer, it shrinks?
Answer:
A balloon kept in sun expands because at high temperature, the Heat energy gained by the particles is transformed into their increased kinetic energy and therefore, they hit the walls of the balloon with greater force exerting more pressure.
When the same balloon is kept in deep freezer because of the low. temperature conditions, the particles lose their kinetic energy and hit the balloon walls with low pressure, making it to contract in size.
Question 27.
Define pressure and atmospheric pressure.
Answer:
Pressure is denned as the thrust acting per emit area. It’s S.I. unit is Pascal but it can also be measured in terms of atmospheric pressure.
The normal atmospheric pressure is the pressure exerted by the air at sea level. It is 760 mm of Hg.
1 Atmospheric pressure (atm) = 1 × 10s Pascal.
Question 28.
How is a vapour different from a gas? Give example.
Answer:
Vapour refers to the gaseous state of a substance that at room temperature exists in its liquid form such as the water vapours formed after the evaporation of liquid water whereas the gas refers to an element or compound that exits in gaseous state even at the room temperature such as oxygen and nitrogen present in air.
Question 29.
What happens when a solid cube of ice is heated in a beaker?
Answer:
When a solid cube of ice is heated, the heat supplied is used up in increasing the vibrations of the particles of the solid but soon the force of attraction reduces enough to allow the particles become free and flow, making the ice to melt.
When the water is continued to be heated, there comes a time when the force of attraction reduces enough to allow particles to leave the water surface and fly free in the atmospheric air in form of vapours.
Question 30.
Define the folio wings:
(i) Latent heat of fusion
(ii) Latent heat of vapourisation
Answer:
(i) Latent heat of fusion: It refers to the heat required to change 1 kg of solid substance into its liquid state at normal atmospheric pressure without any change in temperature at its melting point.
(ii) Latent heat of vapourisation: It refers to the amount of heat required to change 1 kg of liquid substance into its vapours at atmospheric pressure without any change in temperature at normal on its boiling point.
Question 31.
Is the boiling point of water is same in Rajasthan, Shimla, Nanital and Srinagar? Give reason for your answer.
Answer:
No, the boiling point varies with altitude. With the increasing altitude, the boiling point decreases because boiling point is the temperature at which the vapour pressure equals the atmospheric pressure. Hence, as we go at the higher altitudes, the boiling point will decrease with decreasing atmospheric pressure.
Question 32.
On which day, you expect the wet clothes spread on a line to dry faster and why?
(i) Cloudy day or dry day.
(ii) Calm dry day or windy dry day.
Answer:
(i) The wet clothes will dry faster on a dry day as the percentage of water vapours are less in atmospheric air. The atmospheric pressure would be less to oppose the release of vapours from clothes than on a cloudy in which because of the presence of the water vapours, the atmospheric pressure would be more to prevent the release of water vapours.
(ii) On a windy dry day, the clothes will dry faster as the blowing wind would maintain a low pressure over the clothes forcing the wafer vapours to leave the clothes surface.
![]()
Question 33.
Why does evaporation cause the cooling of the surface?
Answer:
Evaporation involves the leaving of the particles of a liquid in gaseous state in air. A particle to leave the surface of the liquid has to have some extra energy. The evaporating particles absorb this required extra energy from the surface producing the cooling effect.
Question 34.
A student kept equal volume of water in same shape and size of the two containers, A of clay and B of plastic at same place for 2 hours. After two hours, water in which container will have lower temperature? Why?
Answer:
In container A, the temperature of water would be less because the clay surface is more porous than plastic hence, from its surface there will be more evaporation of water. The cooling effect of evaporation will reduce more temperature in container A than in container B.
Long Answer Questions
Question 1.
How will you prove that the particles are very-very small beyond our imagination.
Answer:
The matter is made up of very-very tiny particles that are invisible to naked eyes and are even invisible under a microscope but their existence can be proved by the activities like:
- Take 100 ml of water in a beaker and dissolve 2-3 crystals of potassium permanganate.
- Take 10 ml of the above solution and add it to 100 ml of clear water in other beaker.
- Keep diluting the solution like above for 6-8 times.
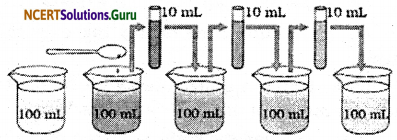
We only observe that the colour only fades with each dilution but does not disappear i.e. the 2-3 crystals of potassium permanganate added to the water initially had millions of the particles that kept distributing themselves with each dilution.
Question 2.
How will you show that gases are more compressible than liquids?
Answer:
To show that the gases undergo compression more easily than liquids, take two 100 ml syringes.
Close their nozzle by inserting them in a rubber cork. Remove the piston, fill the air in one syringe and some water in the other syringe. Place the piston back in its position and apply the pressure.
In syringe filled with air, pressure is applied more easily than in syringe filled with water because, the air, a mixture of gases is more compressible than water.
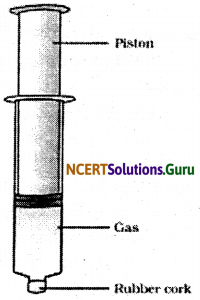
![]()
Question 3.
A student takes a metallic kettle pot partially filled with water and places it over a burner as shown in the figure. What observations will he make when
(a) places the kettle pot on the flame.
(b) after some time of heating over the flame.
(c) he brings a plate filled with ice cubes near the mouth of the kettle’s nozzle. Why?
Answer:
(a) When the kettle is placed over the burner, first kettle body gains heat that is further gained by the water and hence, its temperature starts increasing with increasing time.
(b) After some time, he would see the water vapours in form of steam coming out of the mouth of nozzle.
(c) When he brings a plate filled with ice cubes over the mouth of the nozzle the water vapours present in steam would condense back in liquid water droplets.
Question 4.
What is evaporation? State some its advantages.
Answer:
Evaporation is the loss of water in form of water vapours from the surface of water in a container or a surface at room temperature. The advantage of evaporation include:
- The cooling effect it creates on the surface which is used in rural areas to cool the water and by plants to keep their leaf surface cool.
- The addition of water vapours to atmosphere to regulate the water cycle.
- In loss of solvent in process of crystallization such as extraction of salt from the saline water.
Question 5.
Describe the different factors which affect the rate of evaporation.
Answer:
A number of factors affect the rate of evaporation. These factors include
- Surface area available for evaporation. The rate of evaporation increases with increasing surface area.
- Temperature affects the rate of evaporation as with increasing temperature, the rate of evaporation also increases simultaneously.
- Wind speed, with increasing wind speed, the rate of evaporation also increases. Therefore, the wet clothes dry faster on a hot, dry and windy day.
- Relative humidity, with increasing relative humidity, the rate of evaporation decreases. Therefore, wet clothes dry slower on a humid day than on a dry day.
- Mass of the solvent particles, with the increasing mass, their evaporation decreases.
![]()
Question 6.
Graphically describe the heating curve which a student will plot on heating an ice cube until it gets transformed in state of water vapours.
Answer:
The graph shows that when ice cube is heated, there is no rise in the temperature as the given heat is used up in melting the ice into water. Once in form of water, the temperature starts rising until it reaches 100°C or so. From this temperature onward, he would find no further rise in temperature despite continuous heating as the heat supplied would be used up in transforming the liquid water into its vapours.