These NCERT Solutions for Class 7 Science Chapter 4 Heat Questions and Answers are prepared by our highly skilled subject experts to help students while preparing for their exams.
Heat NCERT Solutions for Class 7 Science Chapter 4
Class 7 Science Chapter 4 Heat Textbook Exercise Questions and Answers
Question 1.
State similarities and differences between the laboratory thermometer and the clinical thermometer.
Answer:
Similarities:
- Both thermometers consist of a long, narrow uniform glass tube.
- Both have a bulb at one end.
- Both contain mercury in bulb.
- Both use Celsius scale on the glass tube.
Differences:
- A clinical thermometer is used to measure human body temperature only while a laboratory thermometer is used to measure temperature of different objects other than human body.
- A clinical thermometer reads temperature between 35°C to 42°C while the range of a laboratory thermometer is -10°C to 110°C.
- Clinical thermometer has a kink near the bulb while there is no kink in a laboratory thermometer.
Question 2.
Give two examples each of conductors and insulators of heat.
Answer:
Conductors – aluminium, iron.
Insulators – plastic, wood.
Question 3.
Fill in the blanks:
a. The hotness of an object is determined by its ……………….
b. Temperature of boiling water cannot be measured by a ………………. thermometer.
c. Temperature is measured in degree ……………….
d. No medium is required for transfer of heat by the process of ……………….
e. A cold steel spoon is dipped in a cup of hot milk. It transfers heat to its other end by the process of ……………….
f. Clothes of ………………. colours absorb heat better than clothes of light colours.
Answer:
(a) temperature
(b) clinical
(c) Celsius
(d) radiation
(e) conduction
(f) dark
Question 4.
Match the following:
| i. Land breeze blows during | (a) Summer |
| ii. Sea breeze blows during | (b) Winter |
| iii. Dark coloured clothes are preferred during | (c) Day |
| iv. Light coloured clothes are preferred during | (d) Night |
Answer:
i. (d)
ii. (c)
iii. (b)
iv. (a)
![]()
Question 5.
Discuss why wearing more layers of clothing during winter keeps us warmer than wearing just one thick piece of clothing.
Answer:
If there is more than one layer of clothes, air would be trapped in between them. Air is a poor conductor of heat. This increases the insulation and thus becomes more comfortable to us by keeping us warm.
Question 6.
Look at figure shown below. Mark where the heat is being transferred by conduction, by convection and by radiation.
Answer:
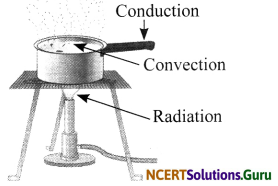
Question 7.
In places of hot climate it is advised that the outer walls of houses be painted white. Explain.
Answer:
Objects with white surface reflect more heat and absorb less. So, less heat from outside would enter the house through walls.
Question 8.
One litre of water at 30°C is mixed with one litre of water at 50°C. The temperature of the mixture will be
(a) 80°C
(b) more than 50°C but less than 80°C
(c) 20°C
(d) between 30°C and 50°C
Answer:
(d) Between 30°C and 50°C.
Question 9.
An iron ball at 40°C is dropped in a mug containing water at 40°C. The heat will
(a) flow from iron ball to water.
(b) not flow from iron ball to water or from water to iron ball.
(c) flow from water to iron ball.
(d) increase the temperature of both.
Answer:
(b) not flow from iron ball to water or from water to iron ball.
Question 10.
A wooden spoon is dipped in a cup of ice-cream. Its other end
(a) becomes cold by the process of conduction.
(b) become cold by the process of convection.
(c) becomes cold by the process of radiation.
(d) does not become cold.
Answer:
(d) does not become cold.
![]()
Question 11.
Stainless steel pans are usually provided with copper bottoms. The reason for this could be that
(a) copper bottom makes the pan more durable.
(b) such pans appear colourful.
(c) copper is a better conductor of heat than the stainless steel.
(d) copper is easier to clean than the stainless steel.
Answer:
(c) copper is better conductor of heat than the stainless steel.
NCERT Extended Learning Activities and Projects
Question 1.
Go to the doctor or your nearest health centre. Observe the doctor taking temperature of patients. Enquire:
a. Why the doctor dips the thermometer in a liquid before use.
b. Why is the thermometer kept under the tongue.
c. Whether the body temperature can be measured by keeping the thermometer at some place other than the mouth.
d. Whether the temperature of different parts of the body is the same or different.
Hint:
a. The doctor dips the thermometer in a liquid so that, the mercury drops to 35 degrees and also so that the infections of the previous person who used the thermometer is also removed.
b. The thermometer is kept under the tongue because the intense heat of our body is present under our tongue.
c. We can measure the temperature of the body by keeping thermometer at some place other than mouth like armpit.
d. The temperature of different parts of the body is not exactly the same but it differs slightly.
Question 2.
Go to a veterinary doctor (a doctor who treats animals). Discuss and find out the normal temperature of domestic animals and birds.
Hint:
Normal body temperature varies greatly between animals. Normal temperature of an animal ranges from 38.3°C to 39.2°C. Normal temperature of a bird is around 40°C.
![]()
Question 3.
Wrap a thin paper strip tightly around an iron rod. Try to burn the paper with candle while rotating the iron rod continuously. Does it burn? Explain your observation.
Hint:
The paper doesn’t bum. The main reason for this consequence is that the iron rod has a high melting point and is a good conductor of heat and hence absorbs the heat and doesn’t allow the paper to reach its ignition temperature.
Question 4.
Take a sheet of paper. Draw a spiral on it as shown in the figure. Cut out the paper along the line. Suspend the paper, as shown in figure, above a lighted candle. Observe what happens. Think of an explanation.

Hint:
As you suspend the spiral paper above the candle. The air around the lighted candle heats up. Since hot air is lighter than cold air, the air that surrounds the candle rises and pushes on your spiral, causing it to spin. The upward movement of hot air causes the movement in spiral paper.
Question 5.
Take two similar transparent glass bottles having wide mouths. Put a few crystals of potassium permanganate or pour a few drops of ink in one bottle. Fill this bottle with hot water. Fill the other bottle with cold water. Cover the cold water bottle with a thick piece of paper such as a postcard. Press the postcard firmly with one hand and hold the bottle with the other hand. Invert the bottle and palace it on top of the hot water bottle. Hold both the bottles firmly. Ask some other person to pull the postcard. Observe what happens. Explain.
Hint:
It can be observed that the, coloured hot water initially ruses up and the clear cold water sinks down at the bottom of the first bottle. Gradually, they mix together to attain a uniform colour and temperature. This demonstrates the process of convection.
Activity 1
Objective: To find the degree of hotness or coldness by the sense of touch.
Materials required: Three bowls, hot water, ice-cold water and water at room temperature.
Procedure:
- Fill the first bowl with the warm water, the second with the ordinary water and the third with the ice-cold water.
- Dip your right hand into the first jar, then dip your left hand into the third jar. Let it be there for one minute and feel the temperature.
- Lift both of your hands each from the first and the third jar, then put it quickly into the second jar.
Observation: During the third step in the activity above, the right hand feels that water is cooler while the left hand feels the water warmer. It means that the touch-sensory organ gives the relative responses to the temperature. Therefore, it is required to use a device to measure the temperature of a body.
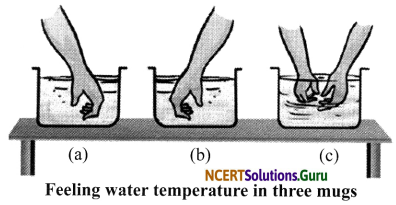
Conclusion: It can be said that our sense of touch fails to give an exact estimation of degree of hotness of a body.
Thermometer: Temperature is measured by a device called thermometer. The thermometer that measures the temperature of our body is called a clinical thermometer.
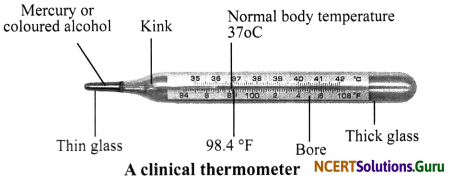
Clinical Thermometer: It consists of a long, narrow, glass the tube with a bulb at – one end filled with mercury. Thermometers have a narrow kink (or constriction) in the fine capillary tube. This constriction prevents the mercury from contracting and flowing back when it is removed from the human body (temperature of surroundings being lower). This enables the recording of the maximum temperature of the body conveniently. The mercury thread needs to be shaken back into the bulb before the thermometer is used again. There is a scale on the thermometer. The scale we use is Celsius scale, indicated by °C. In a clinical thermometer, the range of temperature is form 35°C to 42°C. The normal temperature of the human body is 37°C.
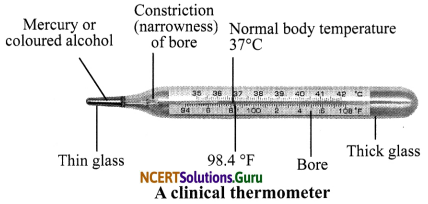
Reading a Thermometer: First of all, note the temperature difference between two bigger marks and also note the number of divisions between these two marks. For example, if the bigger marks read 1°C and there are 10 divisions between them, then one small division in read as \(\frac {1}{10}\) = 0.1°C.
To use it, the thermometer should be washed with an antiseptic. It should be held firmly and given a few jukes to bring the mercury level below 35°C. Its bulb should then be placed under the tongue for one minute before it is taken out to take the reading.
Precautions while using Clinical Thermometer:
- It should be washed with antiseptic before and after use.
- The initial level of mercury should be kept below 35°C.
- It should not be held by its bulb while reading it.
- It should not be kept in sun or near flame.
- It should be handled with care to avoid breakage.
- It should be used to measure human body temperature only.
Laboratory Thermometer: In a laboratory thermometer, the range is form -10°C to 110°C and there is no kink because of which the level of mercury falls rapidly. Such a thermometer can be safely used for measuring the boiling points, melting points or temperatures of substances other than the human body. They are specifically designed for lab purposes.
Precautions while using Laboratory Thermometer: Following are some precautions in addition to those of a clinical thermometer.
- It should be kept upright and not tilted.
- The bulb should be completely immersed in the substance whose temperature is to be measured.
- The bulb should not touch the container walls or base.
Celsius Scale: The Celsius scale to measure temperature was designed by Andres Celsius in 1742. On this scale, temperature is described in degree Celsius. Here, boiling point of water is taken 100°C while melting point of ice is taken 0°C. The other scale which was in use earlier is the Fahrenheit Scale (°F). It has a range of 32°F to 212°F as opposed to the Celsius scale.
Other Types of Thermometers:
i. Maximum- minimum Thermometer:
This thermometer is used to report the maximum and minimum temperatures of previous day reported in weather reports.
ii. Digital Thermometers: Mercury is a toxic substance and is very difficult to dispose off if the thermometer breaks. These days, digital thermometers are available which do not use mercury.

Transfer of heat: The heat always flows from a hotter object to a cooler object. The flow of heat stops when the two objects attain the same temperature. There are three modes of heat transfer. These are: conduction, convection and radiation.
i. Conduction: It is the process by which heat is transferred from the hotter region of an object to the colder region of the same object. Heat transfer in solids generally occurs through conduction.
Conductors: The materials that allow heat to pass through them easily are called conductors of heat. For example, metals like aluminium, iron, and copper are conductors.
Insulators: The materials that do not allow heat to pass through them easily are called poor conductors of heat or insulators. In other words, insulators are the poor conductors of heat. Objects such as wood, plastic, etc., are insulators.
![]()
Activity 2
Objective: To demonstrate the process of conduction.
Materials Required: Five small iron nails, wax, metal rod, burner and a matchbox.
Procedure:
- Fix the heads of the nails on the iron rod with the help of wax at equal distances.
- Arrange the rod as shown in the figure.
- Heat the rod at one end with a burner and observe.
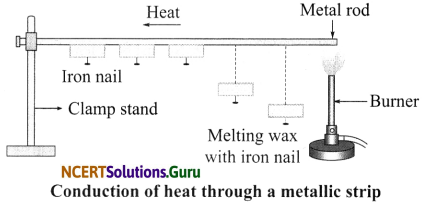
Observation: As heat travels from the left side to the right side along the iron rod, it melts the wax. Due to this, nails fall down one by one.
Conclusion: During conduction, heat is transferred from one particle to the other by means of back and forth vibrations of the particles. There is no actual movement of the particles of the solid from its hotter end to the colder end during the conduction of heat through it.
ii. Convection: It is a type of heat transfer that can only happen in liquids and gases, because it involves movement of particles. Convection happens when there is a difference in temperature between two parts of a liquid or a gas. The hot part of a fluid (liquid or gas) rises up and the cooler part sinks dawn.
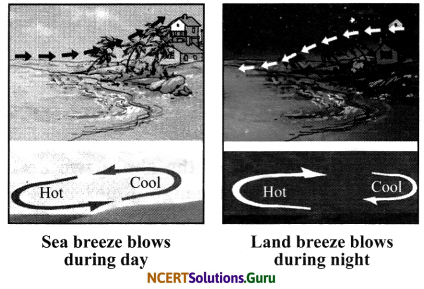
Sea breeze: The flow of cool air from the sea towards the land during the day in coastal areas is called sea breeze. The land gets heated faster than water during the day. Hence, air above land becomes hot and rises up creating a low pressure area. Cool air from water rushes in towards land to fill the space. Hence, windows in coastal areas are made to face the sea to receive the cool sea breeze.

Land breeze: The breeze blowing from the land towards the sea at night is called land breeze. During night, the land cools down faster than the water.
Hence, the hot air over the warm sea rises upwards and the cooler air from the land blows in towards the sea in the form of land breeze.

![]()
Activity 3
Objective: To show the transfer of heat by convection in air.
Materials Required: A candle.
Procedure:
- Light a candle and place it on a stand.
- Now place one hand above the flame (at a safe distance so that you don’t bum your hand) and the other at the side of the flame.

Observation: The hand kept above the flame feels quite hot but the hand kept on the side of the flame does not feel so hot.
Conclusion: The hand above the flame feels hotter since air gets heated by convection and rises up. On the other hand, the air on the side does not feel as hot because there is no convection.
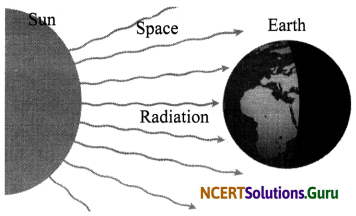
iii. Radiation: It is the process by which heat is transferred without any contact or medium between the heat source and the heated object. We feel the heat from the sun even though we are not touching it. This heat from the sun reaches us by radiation. Thus, transfer of heat by radiation does not require any medium. For example, solar heater traps heat from the sun which can be used for cooking.
Kinds of Clothes we Wear in Summer and Winter: One should wear light coloured clothes, such as white, light pink, light sky blue and other light shades to keep cool because they reflect most of the sun rays back into the atmosphere. Dark fabrics, on the other hand, tend to absorb these rays and trap their heat making one feel even hotter. That is why we feel more comfortable in light-coloured clothes in summers and prefer to wear dark-coloured clothes in winters.
Woollen clothes are not warm but they keep us warm because wool is a poor conductor of heat and it also has air trapped in between the fibres. The trapped air prevents the flow of heat from our body to the cold surroundings as it is a poor conductor of heat. So, we feel warm after wearing woollen clothes in winters.
Nowadays, buildings are also constructed in such a way to trap layers of air by using hallow bricks to prevent heat transfer. This keeps the house cool in summers and warm in winters.
Class 7 Science Chapter 4 Heat Additional Important Questions and Answers
Very Short Answer Type Questions
Question 1.
What is the use of kink in a clinical thermometer?
Answer:
It prevents mercury level from falling on its own when thermometer is removed from the body.
Question 2.
Does transfer of heat by radiation require any medium?
Answer:
The transfer of heat by radiation does not require any medium.
Question 3.
Do all hot bodies radiate heat?
Answer:
Yes, all hot bodies radiate heat.
Question 4.
How does heat transfer take place in liquids and gases?
Answer:
In liquids and gases, heat is transferred by convection.
![]()
Question 5.
What is the name of those thermometers which do not use mercury?
Answer:
Digital thermometers.
Question 6.
Why do we wear woollen clothes during winter?
Answer:
The wool is a poor conductor of heat. Also, it traps air between its fibres which acts as an insulator. Hence, woollen clothes keep us warm during winter.
Question 7.
Name the two types of thermometer.
Answer:
Clinical thermometer and laboratory thermometer.
Question 8.
What is the scale of temperature used in clinical thermometers.
Answer:
Degree Celsius (°C) scale.
Question 9.
What is the average body temperature of a healthy person?
Answer:
37°C.
Question 10.
Which of the two absorbs more radiation – a dark coloured object or a light coloured object?
Answer:
A dark coloured object.
![]()
Question 11.
What are the various methods of transfer of heat?
Answer:
There are three methods of transfer of heat: conduction, convection and radiation
Question 12.
Which type of clothes should be preferred in the month of June in India?
Answer:
Light-coloured clothes.
Question 13.
Which type of cloths should be preferred in the month of February in India?
Answer:
Dark-coloured clothes.
Question 14.
What is a laboratory thermometer?
Answer:
A laboratory thermometer is used for measuring the temperature of substances in a laboratory.
Question 15.
What is the range of a laboratory thermometer?
Answer:
-10°C to 110°C.
Short Answer Type Questions
Question 1.
What is conductor?
Answer:
The materials that allow heat to pass through them easily are called conductors. For example, metals like aluminium, iron and copper.
Question 2.
Define clinical thermometer. Explain its structure with diagram.
Answer:
The temperature is measured by a device called thermometer. The thermometer that measures our body temperature is called clinical thermometer. It consists of a long, narrow glass tube with a bulb fitted at one end. The bulb is filled with mercury. In a clinical thermometer, the range of temperature is form 35°C to 42°C.
Question 3.
What is conduction?
Answer:
Conduction is a process in which transfer of heat takes place between objects by direct contact. In solids generally, the heat is transferred from the hotter end of an object to its cooler end by the process of conduction.
![]()
Question 4.
What is the use of the maximum-minimum thermometer?
Answer:
The maximum and minimum temperature of the atmosphere reported in weather reports is measured by the thermometer called the maximum-minimum thermometer.
Question 5.
What are the different types of thermometers other than clinical and laboratory thermometers?
Answer:
There are two other type of thermometers:
- Maximum- minimum thermometer: This thermometer is used to report maximum-minimum temperature of the day.
- Digital thermometer: Mercury is a toxic substance and is very difficult to dispose off if a thermometer breaks. These days digital thermometers are available which do not use mercury.
Question 6.
Why is conduction only possible in solids?
Answer:
In solids, generally, the heat is transferred by the process of conduction because particles of solids are closely packed and heat is transferred from the hotter end to the colder end of an object. In liquids and gases, the particles are loosely packed and are in motion. Hence, heat transfer by direct contact is difficult.
Question 7.
What is an insulator?
Answer:
The materials that do not allow heat to pass through them easily are called insulators. Insulators are the poor conductors of heat. Materials such as wood, plastic, etc., are insulator.
Question 8.
Why is convection not possible in solids?
Answer:
The molecules of a solid are held strongly and tightly due to intermolecular forces. As these molecules cannot travel to the source of heat energy, convection is not possible in case of solids.
![]()
Question 9.
How do woollen clothes keep us warm in the winter?
Answer:
Woollen clothes are not warm but they keep us warm because wool is a poor conductor of heat and it also has air trapped in between the fibre. The trapped air prevents the flow of heat from our body to the cold surroundings. So, we feel warm after wearing woollen clothes.
Question 10.
What is the precaution that one must follow while handling a clinical thermometer?
Answer:
The following precautions must be taken while handling a clinical thermometer:
- Never hold the thermometer by the bulb while reading it.
- Wash the thermometer with water or antiseptic solution before and after use.
- It should be used to measure the temperature of human body only.
- Read the thermometer by keeping the level of mercury along the line of sight.
Question 11.
Why is mercury preferred in thermometers in place of water?
Answer:
Mercury used due to the following reasons:
- It expands uniformly.
- It does not stick to the walls.
- It is shiny and easy to see.
- It remains liquid at room temperature.
Question 12.
What are the precautions needed to be observed while reading a laboratory thermometer?
Answer:
Following precautions are needed to be observed while reading a laboratory thermometer:
- The thermometer should be kept upright and not tilted.
- Bulb should be surrounded from all sides by the substance of which the temperature is to be measured.
- The bulb should not touch the surface of the container.
Long Answer Type Questions
Question 1.
What are the different modes of heat transfer? Explain.
Answer:
Heat flows from a region of higher to lower temperature till both the bodies attain same temperature. There are three modes of transfer of heat:
- Conduction: Conduction is a process in which transfer of heat takes place between objects by direct contact where heat is transferred from the hotter end of an object to its cooler end. This usually occurs in solids.
- Convection: It is a type of heat transfer that can only happen in liquids and gases because it requires those liquids or gases physically moving. Convection happens when there is a difference in temperature between two parts of a liquid or a gas. The hot part of a fluid rises, and the cooler part sinks.
- Radiation: Radiation is the process by which heat is transferred without any contact between the heat source and the heated object. We feel the heat from the sun even though we are not touching it. This heat from the sun reaches us by radiation. Thus, transfer of heat by radiation does not require any medium.
![]()
Question 2.
Describe with the help of diagrams, the sea breeze and the land breeze.
Answer:
Sea breeze: The flow of cool air from the sea towards the land during the day in coastal areas is called sea breeze. The land gets heated faster than water during the day. Hence, air above the land becomes hot and rises up creating a low pressure area. Cool air from water rushes in towards land to fill the space. Hence, windows in coastal areas are made to face the sea to receive the cool sea breeze.
Land breeze: The breeze blowing from the land towards the sea is called land breeze. During night, the land cools down faster than the water. Hence, the hot air over the warm sea rises upwards and the cooler air from the land blows in towards the sea in the form of land breeze.
Question 3.
Write the key differences between heat and temperature.
Answer:
The differences between heat and temperature can be drawn clearly on the following grounds:
- Heat is nothing but a form of energy in a body. As against this, temperature is something that measures the intensity of heat.
- The main feature of heat is that it travels from a hotter region to a cooler region unlike temperature, which rises when heated and falls when cooled.
- Heat possesses the ability to work but the temperature is used exclusively to gauge the extent of heat.
- The standard unit of measurement of heat is Joules while that of temperature is Kelvin, but it can also be measured in Celsius and Fahrenheit.
- Calorimeter is a device which is used to measure the heat in a body. On the other hand, temperature can be measured by thermometers.
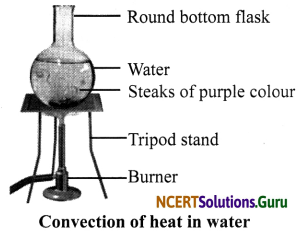
Question 4.
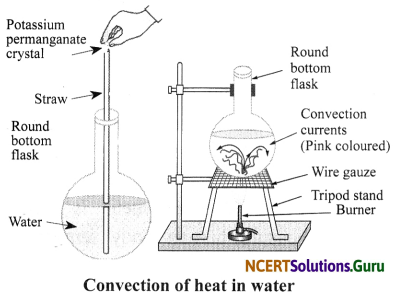
Demonstrate with the help of an activity how heat travels in water?
Answer:
Take a round-bottom flask and fill it with water. Add a few crystals of potassium permanganate to the flask and heat it on a tripod stand as shown below.
As the water gets heated, the hot water starts rising up and the cold water moves towards the flame. This is seen by the movement of purple lines in the water. Also, this process continues till the entire volume of water gets heated. This process of transfer of heat in liquids is called convection.
Picture-Based Questions
Question 1.
Observe the following picture and tell what does this figure show?

Answer:
In the above picture, a girl is reading the temperature in a thermometer. This is a correct method of reading a clinical thermometer.
![]()
Question 2.
What do you observe in the following picture Explain the reason behind it.

Answer:
A metallic cap stuck on a glass bottle can be removed by pouring hot water over the cap. It expands due to a rise in its temperature and can be removed easily.
Question 3.
(a) Observe the following picture and identify (i) and (ii).

(b) Which of the above two occurs during the night time?
Answer:
(a) (i) Sea breeze, (ii) Land breeze.
(b) Sea breeze occurs during daytime and land breeze during night time.