These NCERT Solutions for Class 6 Science Chapter 6 Changes Around Us Questions and Answers are prepared by our highly skilled subject experts to help students while preparing for their exams.
Changes Around Us NCERT Solutions for Class 6 Science Chapter 6
Class 6 Science Chapter 6 Changes Around Us Textbook Exercise Questions and Answers
Question 1.
To walk through a waterlogged area, you usually shorten the length of your dress by folding it. Can this change be reversed?
Answer:
Yes, by unfolding the dress we can reverse this change.
Question 2.
You accidentally dropped your favourite toy and broke it. This is a change you did not want. Can this change be reversed?
Answer:
No, it cannot be reversed. It is an irreversible change.
Question 3.
Some changes are listed in the following table. For each change, write in the blank column, whether the change can be reversed or not.
| S. No. | Change | Can be reversed (Yes/ No) |
| 1. | The sawing of a piece of wood | |
| 2. | The melting of ice candy | |
| 3. | Dissolving sugar in water | |
| 4. | The cooking of food | |
| 5. | The ripening of a mango | |
| 6. | Souring of milk |
Answer:
|
S. No. |
Change |
Can be reversed (Yes/No) |
| 1. | The sawing of a piece of wood | No |
| 2. | The melting of ice candy | Yes |
| 3. | Dissolving sugar in water | Yes |
| 4. | The cooking of food | No |
| 5. | The ripening of a mango | No |
| 6. | Souring of milk | No |
Question 4.
A drawing sheet changes when you draw a picture on it. Can you reverse this change?
Answer:
By erasing the drawing, we can undo the change. For example, if we draw with the help of pencil, we can erase the drawing by using an eraser. Then, it is a reversible change. If we draw using a pen, we cannot erase the drawing. Then this change cannot be reversed.
![]()
Question 5.
Give examples to explain the difference between changes that can or cannot be reversed.
Answer:
Examples of Reversible Changes:
- Melting of ice into water: By freezing the water we can obtain ice again.
- Folding a paper: By unfolding it, we can undo the change.
- Flot milk to cold milk: By boiling milk, we can make it warm again.
Examples of Irreversible Changes:
- Bursting a balloon.
- Bud becoming a flower.
- Cutting a wood into wood pieces.
Question 6.
A thick coating of a paste of Plaster of Paris (POP) is applied over the bandage on a fractured bone. It becomes hard on drying to keep the fractured bone immobilised. Can the change in POP be reversed?
Answer:
When water is added to POP, it undergoes a chemical change. Its composition changes and is converted into another substance. Since it is a chemical change, it cannot be reversed.
Question 7.
A bag of cement lying in the open gets wet due to rain during the night. The next day the sun shines brightly. Do you think the changes, which have occurred in the cement, could be reversed?
Answer:
Due to water, cement hardens and its composition changes. Chemical changes, in general, are irreversible changes. Hence, it is a chemical change which cannot be reversed.
NCERT Extended Learning Activities And Projects
Question 1.
Take a lemon, a paintbrush and a piece of paper. Cut the lemon and squeeze out its juice in a cup. Dip the brush in the lemon juice and write a message on the paper. Let the paper dry and you find that the letters of your message become invisible. Now, press the paper with hot iron or warm it by holding it above the flame of a candle (Take care that it does not catch fire). As the paper gets warm, invisible letters change into dark brown colour. Identify the changes that can be reversed in this process.
Answer:
Irreversible changes: Cutting and squeezing of lemon, writing message on paper.
Reversible changes: Drying and wetting of the paper.
Question 2.
Observe preparation of dishes at your home. Identify two changes that can be reversed.
Answer:
Do it yourself.
Question 3.
Maintain a record for one year of the seasonal changes in vegetables, clothing, nature and events around you. Identify the changes that can or cannot be reversed.
Answer:
Do it yourself.
Activity 1
Objective: To show that a balloon changes its size and shape on blowing air into it and the change is reversible.
Materials Required: A balloon.
Procedure:
- Take a balloon and blow air in it. Take care that it does not burst.
- Now allow the air to escape from the balloon.

Observations: On blowing, the size and shape of the balloon have changed. It regains its shape when air is released.
Conclusion: It is possible to get the balloon back to its original shape and size. Therefore, it is a reversible change.
![]()
Activity 2
Objective: To make a toy aeroplane by folding paper and show that the change is reversible.
Materials Required: A piece of paper.
Procedure:
- Take a piece of paper and fold it to make an aeroplane.
- Now unfold the paper again.
Observation: The sheet of paper has changed into a toy aeroplane. On unfolding, the paper regains original form.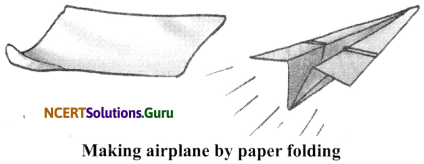
Conclusion: It is a reversible change.
Activity 3
Objective: To show that rolling of roti out of dough is a reversible change.
Materials Required: Dough (kneaded flour) and rolling pin.
Procedure:
- Take a dough and make a small ball of it with hands.
- Roll this ball of dough into a circular roti by using a rolling pin.
- Now, convert this circular roti into small ball again by crumbling.

Observations: The dough rolled into roti was converted back in dough boll.
Conclusion: It is a reversible change.
- Irreversible changes: Changes that cannot be reversed to form the original substance are called irreversible changes. Changing of milk into curd is an irreversible change because curd cannot be turned into milk again.
- Slow changes: Changes which take long time to complete are called slow changes. Growth of plants is a slow change.
- Fast changes: Changes which take short time to complete are called fast changes. For example, burning of candle is a fast change.
- Chemical changes: Changes in which the chemical properties of substances change to form a new substance are called chemical changes. For example, digestion of food.
- Physical changes: Changes in which the physical properties of a substance change and no new substance is formed are called physical changes. For example, melting of candle wax.
- Freezing: Turning of a liquid into solid at low temperature is called freezing. For example, freezing of water into ice.
- Melting: Turning of a solid into liquid when the temperature is high is called melting. For example, melting of ice into water.
- Evaporation: The process of conversion of liquid into vapour at higher temperature is called evaporation. For example, water evaporates in sunlight.
- Condensation: The process of conversion of gas into liquid is called condensation.
- Contraction: A phenomenon in which a substance shrinks or changes its shape is called contraction.
- Expansion: Many substances expand on heating. This property is used to fix metal rim on wooden wheels or wooden handles in certain tools. In such tools, iron blade has a ring in which wooden handle is fitted. The ring is slightly smaller than the handle. It is first heated, due to which it expands and handle is fitted in it easily. On cooling, the shrinks back to original size fitting tightly around the handle.
Class 6 Science Chapter 6 Changes Around Us Additional Important Questions and Answers
Very Short Answer Type Questions
Question 1.
Which type of change ¡s growing of a plant – fast or slow?
Answer:
Slow change.
Question 2.
Do you think that cooking of food is an irreversible change?
Answer:
Yes.
Question 3.
Rolling out roti from dough ball is a slow change or a fast change?
Answer:
Fast change.
![]()
Question 4.
Turning of ice into water and water into ice is a reversible change or irreversible change?
Answer:
Reversible change.
Question 5.
Classify the following into slow and fast changes:
a. Burning of paper
b. Spinning of top
c. Melting of ice cream
d. Change of season
Answer:
a. Fast change
b. Fast change
c. Fast change
d. Slow change
Question 6.
Growth of a plant is an irreversible change. Explain.
Answer:
When a plant becomes a tree, it cannot be reversed back into a plant. So, it is an irreversible change.
Question 7.
What do you think that deforestation is an irreversible change or a reversible change?
Answer:
Irreversible change.
Question 8.
Give an example to show that pressure changes the shape of a material.
Answer:
When an inflated balloon is pressed between two hands, its shape and size change.
![]()
Question 9.
Give an example to show that metal contracts on cooling?
Answer:
An electric wire becomes tight during winters and sag a little during summers. It proves that metal contracts on cooling.
Question 10.
Define: a. evaporation, b. condensation and c. freezing.
Answer:
a. The process of conversion of liquid into vapour is called evaporation.
b. The process of conversion of gas into liquid is called condensation.
c. The change of state from liquid to solid is called freezing.
Question 11.
What are the different ways in which changes can be caused?
Answer:
By mixing, by applying force, by heating and by cooling.
Question 12.
What do you mean by expansion?
Answer:
Expansion is a phenomenon in which a substance expands or changes its shape.
Short Answer Type Questions
Question 1.
Give the characteristics of physical changes.
Answer:
- No new substances are formed.
- Products are identical to reactants in chemical properties.
- These changes are generally reversible.
Question 2.
Give the characteristics of chemical changes.
Answer:
- Properties of product are different from the reactants.
- Most of the chemical changes are irreversible.
- Loss or gain of energy is involved in a chemical change.
![]()
Question 3.
Differentiate between physical changes and chemical changes with examples.
Physical change
Answer:
| Physical change | Chemical change |
| 1. The change in which only the physical properties of a substance are changed and no new substance is formed is called a physical change. | 1. The change in which chemical reactions take place and a new substance is formed, is called a chemical change. |
| 2. This is a reversible change. | 2. Most of these changes are irreversible. |
| 3. Example: melting of ice and freezing of water. | 3. Example: change of milk into curd. |
Question 4.
Explosion of cracker is an irreversible change. How?
Answer:
Explosion of a cracker is an irreversible change because once crackers are burnt, they cannot be converted back to their original form.
Question 5.
How candles of different shapes are made?
Answer:
Molten wax is poured in desired mould and a wick is placed in it. Then it is cooled. This is how we get candles of different shapes.
![]()
Question 6.
State a few examples each for slow changes and fast changes.
Answer:
Examples of slow changes:
- Germination of seed
- Rusting of iron
- Curdling of milk
Examples of fast changes:
- Burning of candle
- Burning of paper
- Inflating a tire
Question 7.
Formation of clouds is usually a physical change. Justify the statement.
Answer:
The formation of clouds is a physical change because there is a phase transformation cycle of natural water from liquid to gas and then, gas to liquid. Hence, the property of water never changes.
Question 8.
How do we convert milk into curd and what type of change is it?
Answer:
The warm milk is taken in a vessel and a small quantity of curd is added into it. The milk is stirred and it is kept aside for few hours without any disturbance. Finally, the milk is converted into curd. However, curd formed cannot be converted into milk again indicating that it is an irreversible change.
Question 9.
What will be the change when sugar is heated?
Answer:
When sugar is continuously heated, a black powdery substance is formed which is called charcoal. Since a new product is formed, it is termed to be a chemical change.
Question 10.
Rolling of roti from dough is a reversible change. Explain.
Answer:
Take some dough and make a ball of it. Roll this ball of dough into a circular shape by using a rolling pin. Now, if we want, we can convert this rolled roti back into the ball of dough. This proves that the rolling of roti out of dough is a reversible change.
Long Answer Type Questions
Question 1.
Why making statues out of plaster of Paris is an irreversible and chemical change?
Answer:
Making statues out of plaster of Paris is an irreversible and chemical change because Plaster of Paris has a property of setting into a hard mass on getting mixed with water due to the formation of a new compound. The hard mass cannot be converted back into the previous plaster of Paris. Hence, it is an irreversible chemical change.
Question 2.
How is inflating a balloon different from bursting an inflated balloon?
Answer:
Inflating a balloon is an example of a reversible change whereas bursting a balloon is an irreversible change. When we blow air into a balloon, its shape and size changes. But it regains its original form when the air is allowed to escape. On the other hand, a burst balloon cannot be changed back to the original form.
![]()
Question 3.
What are desirable and undesirable changes? Explain with examples.
Answer:
Desirable changes: Those changes which are desired to occur, that is, we want them to take place are known as desirable changes. Example: ripening of fruits, germination of seeds, etc.
Undesirable changes: Those changes which we do not want to take place are called undesirable changes. Example: rusting of iron, souring of milk, rotting of fruits, etc.
Question 4.
The burning of paper is said to be an irreversible change whereas the boiling of water is known as reversible change. Give reason.
Answer:
The paper converts into ash on burning which we cannot combine to form the original piece of paper again. Therefore, burning of paper is said to be an irreversible change. On the other hand when we boil water by heating, then it changes into steam. On cooling, the steam, it again converts into water by condensation which is a reversible change. Thus, burning of paper is said to be an irreversible change whereas the boiling of water is known as reversible change.
Question 5.
When is a change said to have taken place in a material? Explain with the help of an example.
Answer:
The things around us have certain properties such as state (solid, liquid, gas), position, shape, size, colour, temperature, composition and structure, etc. When one or more properties of a thing become different, the changes in it have taken place. Changes involve different kinds of alterations in the things. When a change takes place, there may be change in the state, position, shape, size, colour, temperature, composition or structure of the material of the object. Example: when ice melts, it forms water. Ice is a solid, whereas water is a liquid. So, the melting of ice involves a change in state (from solid to liquid state).
Picture-Based Questions
Question 1.
Observe the given pictures and answer the following questions:
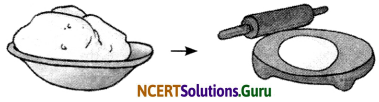
a. What type of change it is – physical or chemical?
b. Can this change be reversed?
c. Can this change be reversed after roti is cooked on flame?
Answer:
a. Physical change
b. Yes
c. No
![]()
Question 2.
Observe the given picture and answer the questions that follow:

a. Name the process by which above change takes place.
b. Name the process by which this change can be reversed.
Answer:
a. Melting
b. Freezing