Students may want to download NCERT MCQ Questions for Class 12 Chemistry Chapter 11 Alcohols, Phenols and Ethers with Answers Pdf free download. These Alcohols, Phenols and Ethers Class 12 MCQs Questions with Answers can help understand the concepts score better in your Class 12 Chemistry Exam, so make sure you practice these! Check out chapter-wise MCQ Questions for Class 12 Chemistry with Answers.
Alcohols, Phenols and Ethers Class 12 MCQs Questions with Answers
Solve these Alcohols, Phenols and Ethers Multiple Choice Questions of Class 12 Chemistry Chapter 11 MCQ, so as not to miss out on any concept from being clear about what they mean!
Question 1.
Dehydration of tertiary alcohols with Cu at 573 gives
(a) Aldehydes
(b) Ketones
(c) Alkenes
(d) None of these
Answer
Answer: (c) Alkenes
Question 2.
Molecular formula of ethers is:
(a) CnH2n+2O
(b) CnH2nO
(c) CnH2n+1O
(d) None of these
Answer
Answer: (a) CnH2n+2O
Question 3.
Williamson’s synthesis is an example of:
(a) Nucleophilic substitution reaction
(b) Nucleophilic addition
(c) Electrophilic substitution
(d) None of the above
Answer
Answer: (a) Nucleophilic substitution reaction
Question 4.
Reaction used for the preparation of ethers is
(a) Reimer-Tiemann reaction
(b) Williamson’s synthesis
(c) Wurtz reaction
(d) Cannizzaro reaction
Answer
Answer: (b) Williamson’s synthesis
Question 5.
The test used to distinguish alcohols from one another is known as
(a) Hinsberg’s test
(b) 2, 4-DNP test
(c) Iodoform test
(d) Lucas Lest
Answer
Answer: (d) Lucas Lest
Question 6.
The IUPAC name of
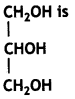
(a) Propane-1, 3-diol
(b) Propane-1, 2-diol
(c) Propane -1, 2, 3-triol
(d) Glycerol
Answer
Answer: (c) Propane -1, 2, 3-triol
Question 7.
Ethers on hydrolysis give
(a) carboxylic acid
(b) alcohol
(c) ester
(d) ketone
Answer
Answer: (b) alcohol
Question 8.
Which of the following has highest boiling point?
(a) Methanol
(b) Ethanol
(c) Propan-1 -ol
(d) Butan-1-ol
Answer
Answer: (d) Butan-1-ol
Question 9.
Which has highest value of pKa?
(a) Phenol
(b) Ethanol
(c) o-Nitrophenol
(d) o-Cresol
Answer
Answer: (b) Ethanol
Question 10.
Which of the following is most acidic?
(a) Benzyl alcohol
(b) Cyclohexanol
(c) Phenol
(d) m-Chlorophenol.
Answer
Answer: (d) m-Chlorophenol.
Question 11.
Phenols on reaction with cone. HNO3 in the presence of cone. H2SO4 give
(a) o-nitrophenol
(b) m-nitrophenol
(c) p-nitrophenol
(d) 2,4,6-trinitrophenol.
Answer
Answer: (d) 2,4,6-trinitrophenol.
Question 12.
Methanol and ethanol can be distinguished by using
(a) Fehling’s test
(b) Iodoform test
(c) Tollen’s test
(d) Carbylamine test.
Answer
Answer: (b) Iodoform test
Question 13.
The IUPAC name of the compound
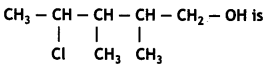
(a) 2-Chloro-3,4-dimethyl-n-pentyl alcohol
(b) 2-Chloro-3,4-dimethylpentan-5-ol
(c) 4-Chloro-2,3-dimethylpentan-1 -ol
(d) 2,3-Dimethyl-4-chloropentan-1 -ol.
Answer
Answer: (c) 4-Chloro-2,3-dimethylpentan-1 -ol
Question 14.
![]()
A will be
(a) CH2=CH2
(b) C2H5OCH3
(c) (C2H5)2O
(d) CH3CH2CH2CH3
Answer
Answer: (c) (C2H5)2O
Question 15.
Which is weakest acid in the following?
(a) CH3OH
(b) (CH3)2CHOH
(c) CH3CH2OH
(d) (CH3)3COH
Answer
Answer: (a) CH3OH
Question 16.
Williamson synthesis is used to prepare:
(a) Alcohol
(b) Amine
(c) Ketone
(d) Ether
Answer
Answer: (d) Ether
Question 17.
In the reaction
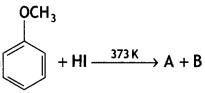
A and B are:
(a) C6H5l, CH3OH
(b) C6H5OH, CH3l
(c) C6H5CH2OH, CH3l
(d) CH3CH2l, C6H5OH
Answer
Answer: (b) C6H5OH, CH3l
Question 18.
The IUPAC name of
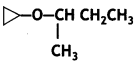
(a) 2-Cyclopropoxybutane
(b) 2-Propoxybutane
(c) 2-Propoxypropane
(d) 2-Methyl-2-propoxypropane
Answer
Answer: (a) 2-Cyclopropoxybutane
Question 19.
Neutral ferric chloride test can be used to distinguish between
(a) Alcohols and ethers
(b) Aldehydes and ketones
(c) Amines and aldehyde
(d) Phenols and alcohols
Answer
Answer: (d) Phenols and alcohols
Question 20.
On reaction with aqueous bromine at room temperature, phenol forms which of the following?
(a) meta-Bromophenol
(b) 2, 6-Dibromophenol
(c) 2, 4, 6-Tribromophenol
(d) 3, 5-Dibromophenol
Answer
Answer: (c) 2, 4, 6-Tribromophenol
Question 21.
How many alcohols with molecular formula C4H10O are chiral in nature?
(a) 1
(b) 2
(c) 3
(d) 4
Answer
Answer: (a) 1
Question 22.
What is the correct order of reactivity of alcohols in the following reaction?
![]()
(a) 1° > 2° > 3°
(b) 1° < 2° > 3°
(c) 3° > 2° > 1°
(d) 3° > 1° > 2°
Answer
Answer: (c) 3° > 2° > 1°
Question 23.
CH3CH2OH can be converted into CH3CHO by
(а) catalytic hydrogenation
(b) treatment with LiAlH4
(c) treatment with pyridinium chlorochromate
(d) treatment with KMnO4
Answer
Answer: (c) treatment with pyridinium chlorochromate
Question 24.
The process of converting alkyl halides into alcohols involves
(a) addition reaction
(b) substitution reaction
(c) dehydrohalogenation reaction
(d) rearrangement reaction
Answer
Answer: (b) substitution reaction
Question 25.
Which of the following compounds is aromatic alcohol?
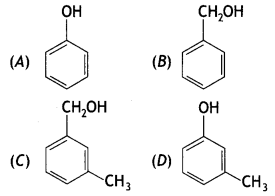
(a) A, B, C, D
(b) A, D
(c) B, C
(d) A
Answer
Answer: (c) B, C
Question 26.
Give IUPAC name of the compound given below.
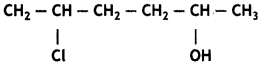
(a) 2-Chloro-5-hydroxyhexane
(b) 2-Hydroxy-5-chlorohexane
(c) 5-Chlorohexan-2-ol
(d) 2-Chlorohexan-5-ol
Answer
Answer: (c) 5-Chlorohexan-2-ol
Question 27.
IUPAC name of m-cresol is
(a) 3-methylphenol
(b) 3-chlorophenol
(c) 3-methoxyphenol
(d) benzene-1,3-diol
Answer
Answer: (a) 3-methylphenol
Question 28.
IUPAC name of the compound
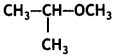
(a) 1-methoxy-1-methylethane
(b) 2-methoxy-2-methylethane
(c) 2-methoxypropane
(d) isopropylmethyl ether
Answer
Answer: (c) 2-methoxypropane
Question 29.
Which of the following species can act as the strongest base?
(a) ΘOH
(b) ΘOR
(c) ΘOC6H5
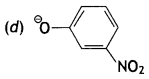
Answer
Answer: (b) ΘOR
Question 30.
Which of the following compounds will react with sodium hydroxide solution in water?
(a) C6H5OH
(b) C6H5CH2OH
(c) (CH3)3COH
(d) C2H5OH
Answer
Answer: (a) C6H5OH
Question 31.
Phenol is less acidic than
(a) ethanol
(b) o-nitrophenol
(c) o-methylphenol
(d) o-methoxyphenol
Answer
Answer: (d) o-methoxyphenol
Question 32.
Which of the following is most acidic?
(a) Benzyl alcohol
(b) Cyclohexanol
(c) Phenol
(d) m-Chlorophenol
Answer
Answer: (d) m-Chlorophenol
Question 33.
Mark the correct order of decreasing acid strength of the following compounds.
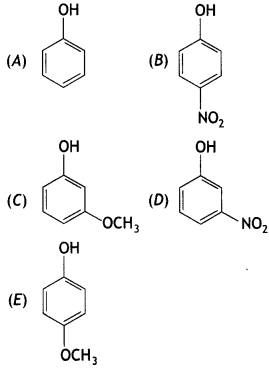
(a) E>D>B>A>C
(b) B>D>A>C>E
(c) D>E>C>B>A
(d) E>D>C>B>A
Answer
Answer: (b) B>D>A>C>E
Question 34.
Mark the correct increasing order of reactivity of the following compounds with
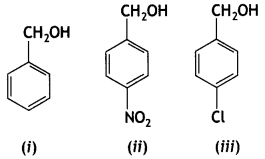
(a) (i) < (ii) < (iii)
(b) (ii) < (i) < (iii)
(c) (ii) < (iii) < (i)
(d) (iii) < (ii) < (i)
Answer
Answer: (c) (ii) < (iii) < (i)
Question 35.
Arrange the following compounds in increasing order of boiling point.
Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
(a) Propan-1-ol, butan-2-ol, butan-1-ol, pentan-1-ol
(b) Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
(c) Pentan-1-ol, butan-2-ol, butan-1-ol, propan-1 -ol
(d) Pentan-1-ol, butan-1-ol, butan-2-ol, propan-1 -ol
Answer
Answer: (a) Propan-1-ol, butan-2-ol, butan-1-ol, pentan-1-ol
Assertion and Reason Type Questions
The questions given below consist of an assertion and a reason. Use the following key to choose the appropriate answer.
(a) Both Assertion (A) and Reason (R) are correct statements, and Reason (R) is the correct explanation of the Assertion (A).
(b) Both Assertion (A) and Reason (R) are correct statements, but Reason (R) is not the correct explanation of the Assertion (A).
(c) Assertion (A) is correct, Reason (R) is wrong statement.
(d) Assertion (A) is wrong, but Reason (R) is correct statement.
Question 36.
Assertion: C2H5OH is a weaker base than phenol but is a stronger nucleophile than phenol.
Reason: In phenol the lone pair of electrons on oxygen is withdrawn towards the ring due to resonance.
Answer
Answer: (d) Assertion (A) is wrong, but Reason (R) is correct statement.
Question 37.
Assertion: The boiling points of alcohols are higher than those of hydrocarbons of comparable molecular mass.
Reason: Alcohols show intramolecular hydrogen bonding.
Answer
Answer: (c) Assertion (A) is correct, Reason (R) is wrong statement.
Question 38.
Assertion: Phenol undergoes Kolbe’s reaction but ethanol does not.
Reason: Phenol is more acidic than ethanol.
Answer
Answer: (b) Both Assertion (A) and Reason (R) are correct statements, but Reason (R) is not the correct explanation of the Assertion (A).
Question 39.
Assertion: The C-O-C bond angle in ethers is higher than H-O-H bond angle in water.
Reason: Oxygen in both ethers and water is sp³ hybridised.
Answer
Answer: (b) Both Assertion (A) and Reason (R) are correct statements, but Reason (R) is not the correct explanation of the Assertion (A).
Question 40.
Assertion: The boiling point of diethyl ether is much less than that of ethanol.
Reason: In ethanol, the molecules are associated by the formation of intermolecular hydrogen bonding, whereas in diethyl ether, it is not possible.
Answer
Answer: (a) Both Assertion (A) and Reason (R) are correct statements, and Reason (R) is the correct explanation of the Assertion (A).
Question 41.
Assertion: Alcohols have higher boiling points than ethers of comparable molecular masses.
Reason: Alcohols and ethers are isomeric compounds.
Answer
Answer: (b) Both Assertion (A) and Reason (R) are correct statements, but Reason (R) is not the correct explanation of the Assertion (A).
Question 42.
Assertion: o- and p-nitrophenols can be separated by steam distillation.
Reason: o-isomer is steam volatile due to chelation, while p-isomer is not steam volatile due to association of molecules by intermolecular H-bonding.
Answer
Answer: (a) Both Assertion (A) and Reason (R) are correct statements, and Reason (R) is the correct explanation of the Assertion (A).
This NCERT MCQ Questions for Class 12 Chemistry Chapter 11 Alcohols, Phenols and Ethers with Answers Pdf free download has been put together to help students understand the CBSE Class 12 Chemistry Alcohols, Phenols and Ethers MCQs. Hope you found this helpful!