CBSE Sample Papers for Class 10 Science paper 2
These Sample papers are part of CBSE Sample Papers for Class 10 Science. Here we have given CBSE Sample Papers for Class 10 Science Paper 2
Time allowed: 3 hours
Maximum marks : 80
General Instructions
- The question paper comprises two sections, A and B. You are to attempt both the sections.
- All questions are compulsory.
- All questions of Section-A and B are to be attempted separately.
- There is an internal choice in two questions of three marks each and one question of five marks.
- Question numbers 1 and 2 in Section-A are one mark question. They are to be answered in one word or in one sentence.
- Question numbers 3 to 5 in Section-A are two marks questions. These are to be answered in 30 words each.
- Question numbers 6 to 15 in Section-A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section-A are 5 mark questions. These are to be answered in 70 words each.
- Question numbers 22 to 27 in Section-B are based on practical skills. Each question is a two marks question. These are to be answered in brief.
SECTION-A
Question 1.
In which organisms can we see reproduction by regeneration ? [1]
Question 2.
Which part of the nervous system controls reflex actions ? [1]
Question 3.
Why are covalent compounds poor conductors of electricity ? [2]
Question 4.
Name the type of mirrors used in the design of solar furnaces. Explain how high temperature is achieved by this device. [2]
Question 5.
What is fossils. [2]
Question 6.
What kind of eye defects are corrected by bi-focal lenses ? [3]
OR
A concave mirror produces three times magnified (enlarged) real image of object placed at 10 cm in front of it. Where is the image located ?
Question 7.
With the help of a diagram of experimental setup describe an activity to show that the force acting on a current carrying conductor placed in a uniform magnetic field increases with increase in field strength. [3]
Question 8.
Why do we apply paint on iron particles ? [3]
Question 9.
Why is sodium hydrogen carbonate used in antacids ? [3]
Question 10.
Explain the ways in which glucose is broken down in the absence of oxygen. [3]
OR
Describe the role of alveoli in respiration in human beings.
Question 11.
“Evolution and classification of organisms are interlinked.” Give reasons to justify this statement. [3]
Question 12.
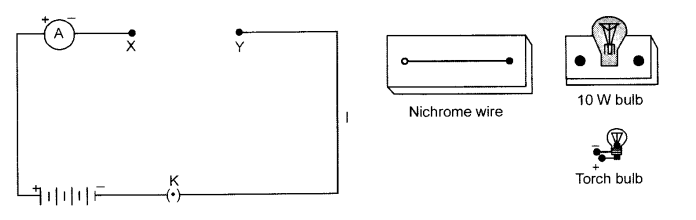
Set up a circuit by connecting four dry cells of 1.5 V each in series with the ammeter leaving a gap XY in the circuit as shown below : [3]
Complete the circuit by connecting the nichrome wire in the gap XY. Plug the key. Note the ammeter reading. Remove the key from the plug. Replace the nichrome wire with torch bulb and then a 10 W bulb. Read the ammeter in each case. What are your observations ?
Question 13.
Distinguish between saponification and esterification reaction using chemical equations. State one use each of esters and saponification process. [3]
OR
What happens when:
(a) Ethanol is burnt in air ?
(b) Ethanol is heated with excess cone. H2S04 at 443 K.
(c) A piece of sodium is dropped into ethanol.
Explain with chemical equations.
Question 14.
What is watershed management ? [3]
Question 15.
What are the advantages and disadvantages of using a solar cooker ? [3]
Question 16.
What are the factors on which resistance of a conductor depends ? Explain with an activity. [5]
Question 17.
(a) What effect does the concentration of H+(aq) ions have on the nature of a solution ? [5]
(b) What effect does the concentration of H– ions have on the nature of a solution ?
(c) Someone put some universal indicator paper into vinegar. The pH is 3. What does this tell you about the vinegar ?
(d) Someone put some universal indicator paper onto wet soap. The pH is 8. What does this tell you about the soap ?
(e) State whether a solution is acidic, alkaline or neutral if its pH is :
- 9
- 4
- 7
- 1
- 10
- 3
OR
(a) Define indicator. Name two indicators obtained from plants.
(b) Write balanced chemical equation for the reaction that takes place when sodium oxide reacts with water. How will this solution behave towards phenolphthalein and red litmus paper ?
(c) State what happen when sodium hydroxide solution reacts with dilute hydrochloric acid. What is this reaction called ?
Question 18.
Describe the properties of ethanol. [5]
Question 19.
Show the refraction of light through a glass slab using a ray diagram. What is refractive
index ? [5]
Question 20.
What is the difference between soaps and detergents ? What is the mechanism of the cleansing action of soaps ? Why don’t soaps form lather with hard water ? Mention any two problems that arise on using detergents instead of soap. [5]
Question 21.
What changes would you suggest in your home in order to be environment friendly ? [5]
OR
Describe the energy flow in a food chain.
SECTION-B
Question 22.
You have four test tubes A, B, C and D containing sodium carbonate, sodium chloride, lime water and blue litmus solutions respectively. Of these four which test tube material will give the correct test for acetic or ethanoic acid ? [2]
(a)Only A
(b) A and B
(c) B and C
(d) A and D
Question 23.
A gas is liberated immediately with brisk effervescence when acetic acid is added to sodium hydrogen carbonate powder in a test tube. Name the gas and describe the test that confirms the identity of the gas. [2]
Question 24.
Select the set of homologous organs from the following: [2]
(a) Wings of pigeon and butterfly
(b) Wings of pigeon and bat
(c) Wings of butterfly and bat
(d) Forelimbs of a dog, frog and crocodile.
Question 25.
Tina covered a leaf from a de-starched plant with a black strip of paper and kept it in the garden outside her house in fresh air. In the evening she tested the covered portion for starch. Why did she do so ? [2]
(a) To prove that C02 is given out during respiration
(b) To prove that C02 is necessary for photosynthesis
(c) To prove that chlorophyll is necessary for photosynthesis
(d) To prove that light is necessary for photosynthesis.
Question 26.
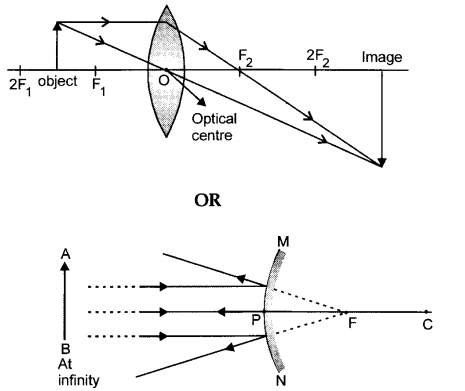
Draw a labelled ray diagram to show the formation of an image by a convex lens when an object is placed in front of the lens between F1 and 2F1. [2]
OR
Draw a labelled ray diagram to show the formation of image in a convex mirror when the object is at infinity. Mark clearly the pole and focus of the mirror in the diagram.
Question 27.
In an experiment, to study the dependence of potential difference (V) on the electric current (I) across a conductor (resistor), if the circuit is on for a long time, which of the following statements are correct ? [2]
(a) Zero error of the ammeter changes.
(b) Zero error of the voltmeter changes.
(c) Value of resistance changes.
(d) Resistor gets heated.
ANSWERS
SECTION-A
Answer 1.
Simple animals like Planaria and Hydra, when cut up into pieces, each piece can grow into a new and complete organism. This process is called regeneration. Regeneration is carried out by specialised cells. These cells proliferate and give rise to a large number of cells which in turn undergo specialisation to form the complete organism.
Answer 2.
Reflex action means actions that take place in a fraction of a second. Some examples of reflex action are with drawing of the hand quickly when it comes in contact with something hot or sharp, withdrawing the feet when one steps on a sharp object. Since these actions occur very fast they are controlled by the spinal cord. Receiving the message and quickly deciding on the right action is done by the spinal cord. The brain is not involved here.
Answer 3.
Covalent compounds are formed by the sharing of electrons between the participating elements. Elements like carbon have tetravalency meaning four electrons in their outermost shell. In order to get the stable gas configuration they need another four electrons. So they will bond with another element which can share the electron with them.
For example in the formation of carbon tetrachloride, a carbon atom will combine with four chloride atoms. There is a single bond formed with all four chloride atoms. Now, the carbon has eight electrons in its outermost shell and the chloride atoms too have eight electrons in their outermost shell. Thus, the atoms are held by strong forces of attraction between them. There are no ions formed and hence they are poor conductors of electricity.
Answer 4.
The mirror used in the design of solar furnaces is the concave mirror. When the solar furnace is placed at the focus of a large concave mirror called reflectors it focuses a parallel beam of light on the furnace. This results in the high temperature.
Answer 5.
Fossils are the remains or the preserved traces of organisms that existed a long time back. Sometimes when organisms die, their body parts are not totally decomposed, some part may get preserved in an environment (mud or layers of rock particles) that does not destroy it completely. After many years either these body parts or their impressions are discovered and become important clues in support of evolution.
Answer 6.
The human eye is a wonderful organ which helps us to see the colourful world around us. In front of the eye we have a crystalline lens that provides the finer adjustment of focal length required to focus objects at different distances on the retina. The eye lens forms an inverted real image of the object on the retina.
With age the power of accommodation of the eye decreases. For most people, the near point gradually recedes away. They find it difficult to see nearby objects comfortably and distinctly without corrective eye glasses. This defect is called far-sightedness or presbyopia. It is caused by the gradual weakening of the ciliary muscles and diminishing flexibility of the eye-lens. Sometimes a person may suffer from both myopia and hypermetropia. Such people require bifocal lenses. A common type of bifocal lens consists of both concave and convex lens. The upper part is the concave lens for distant vision and the lower part is convex lens for near vision. Nowadays it is possible to correct refractive defects with contact lenses or through surgical processes.
OR
Let the height of the object = h
Height of the image, h’ = – 3h (image formed is real)
Object distance, u = -10 cm
Then, magnification produced by a spherical mirror is given by the relation,
\(m=\frac { { h }^{ ‘ } }{ h } =\frac { -\nu }{ u } \)
Substituting the values, we get
\(-\frac { 3h }{ h } =-\frac { v }{ u } \)
\(-3\quad =\quad \frac { -\nu }{ 10 }\)
υ = -30 cm
Here, the negative sign indicates that an inverted image is formed at a distance of 30 cm in front of the given concave mirror.
Answer 7.
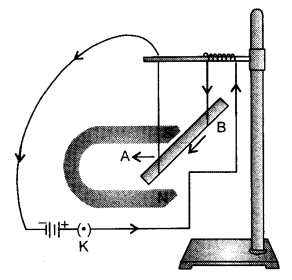
Take a battery, two magnets of different sizes and intensities, small aluminium rod AB, two connecting 1 wires and an iron stand and arrange them as shown in the figure above. Now bring the magnet with lower magnetic intensity closer to the aluminium rod. The rod gets deflected. Now observe the velocity of the rod. The displacement of the rod in the above activity suggests that a force is exerted on the
current carrying conductor when it is placed in a magnetic field.
Now steady the rod and bring both the magnets closer together to ensure that the magnetic field is stronger than before. The rod moves towards the magnet but faster than before. This is possible only if the force acting on the rod is stronger than before and because the body will be starting with zero velocity it will move faster only if the acceleration is more. Since acceleration is more, the force acting on it is more. Thus, the force acting on a current carrying conductor placed in a uniform magnetic field increases with increase in the field strength.
Answer 8.
Paint is applied on iron articles to create a coating on the iron surface and prevent the reaction of iron with-atmospheric oxygen. Usually iron and even other metals like copper and silver undergo oxidation when exposed to atmosphere for a long time. Iron articles are shiny when new but get coated with a reddish brown powder when exposed for some time. This is commonly called rusting of iron. When the metals like iron, copper and silver are exposed to moisture and acids over a period of time they get corroded and the process is called corrosion. These reactions damage the articles and weaken them, Corrosion of iron is a serious problem. Vehicles, bridges, ships and buildings all use iron as their structure and if this gets corroded it weakens the whole thing. Hence, every year a lot of money is
spent repairing these damaged parts. It might be sensible to give these things a regular coating of paint and keep them free of moisture and dust as far as possible to save a lot of money.
Answer 9.
Sodium hydrogen carbonate is used in antacids because it is a mild non-corrosive base. It neutralises the acidity of hydrochloric acid found in the stomach. Usually it is given to patients with acidity as milk of magnesia (Magnesium hydroxide).
Sodium hydrogen carbonate is the chemical name of the compound baking soda. It is widely used in kitchens as an ingredient to make fluffy and crispy food items.
It is produced by using sodium chloride as one of the raw materials:
NaCl + H2O + CO2 + NH3 → NH4Cl + NaHCO3
The following reaction takes place when it is heated while cooking.
2NaHCO3 \(\underrightarrow { heat }\) Na2CO3 + H2O + CO2
The C02 causes bread or cake to rise making them spongy and soft.
Answer 10.
The first step in respiration is the breakdown of glucose, a six-carbon compound into a three-carbon compound called pyruvate. This process takes place in the cytoplasm. Further, the pyruvate may be converted into ethanol and carbon dioxide. This process takes place in yeast during fermentation. As this process takes place in the absence of air (oxygen) it is called anaerobic respiration. The release of energy during anaerobic respiration is lesser than during aerobic respiration. The energy released during respiration is used to synthesise ATP-Adenosine Tri-Phosphate, the energy currency of the cell. The ATP supplies energy to carry out different endothermic reaction taking place in the cell.
Glucose (6-C molecule) → Pyruvate (3-C molecule) + Energy → Absence of oxygen in yeast → Ethanol (2-C molecule) + CO2 + energy
OR
In human beings, air enters the body through a pair of nostrils. Fine hairs in the nostrils filter the air. Mucous in the passage of the nose help in this process. From here air enters the throat and then moves into the lungs. The passage in the throat is the trachea which is lined by cartilaginous rings. Within the lungs the passage divides into finer and finer tubes leading into small balloon-like structures called alveoli. The alveoli have very thin walls richly supplied with blood capillaries where the exchange of gases takes place. When air is breathed in, the chest cavity expands and air is sucked into the lungs and fill in the expanded alveoli. Blood brings in carbon dioxide from the rest of the body to be released into the alveoli. In exchange, oxygen in the alveolar air is taken up by the blood in the alveolar vessels to be transported to all the cells in the body. This is the role of the alveoli in respiration in human beings.
Answer 11.
Classification is the grouping and naming of organisms based on the similarities and differences in their characteristics. Organisms are classified based on their inherited and acquired characteristics. Hence as we move on from simple to complex organisms, we are moving along the path of evolution. Evolution means the slow process of change from simple to complex form of organisms. All living forms on the earth have undergone evolution. So the hierarchy of classification reveals how evolution could have taken place and the evolutionary relationship among organisms. Classification also helps to predict and identify common ancestors.
Answer 12.
The observations based on the ammeter readings are as follows:
- The current reading is different for the different components. This is because certain components offer an easy path for the flow of electric current while the others resist the flow.
- It is known that the motion of electrons in an electric circuit constitutes an electric current. The electrons however are not completely free to move within a conductor.
- The electrons are restrained by the attraction of the atoms among which they move. Hence, the motion of electrons through a conductor is retarded by its resistance.
- A component of a given size that offers a low resistance is a good conductor.
- An insulator offering sufficient resistance is called a resistor.
- A component of identical size that offers a high resistance is called a poor conductor.
Answer 13.
In saponification, esters react in the presence of an acid or a base to give back the alcohol and carboxylic acid.
In saponification, RCOOR’ + NaOH → RCOONa + R’OH
Esterification is opposite of saponification. Esters are most commonly formed by reaction of an acid and an alcohol.
In esterification, RCOOH +R’OH \(\underrightarrow { heat }\) RCOOR’+ H2O
Uses of esters:
(i) They are sweet smelling substances.
(ii) They are used in making perfumes and as flavouring agents.
Uses of saponification reaction:
It is used to manufacture soap and soap products on a large scale.
OR
(a) CH3CH2OH + 3O2 \(\underrightarrow { air }\) 2C02 + 3H2O + heat + light
(b) CH3CH2OH \(\xrightarrow [ 443K ]{ conc.{ H }_{ 2 }S{ O }_{ 4 } } \) CH2 = CH2 + H2O
(c) 2CH3CH2OH + 2Na → 2CH3CH2OH + H2
Answer 14.
Watershed management: Water happens to be an important resource for all the terrestrial forms of life on this earth. Water is a resource whose demand will go on increasing due to the manifold increase in the number of all the living things on this earth. Water plays an important role as a resource and as a part of the water cycle. Human intervention has polluted this water and affected the availability of water in various regions.
Watershed management emphasises scientific soil and water conservation in order to increase the biomass production. The aim is to develop primary resources of land and water to produce secondary resources of plants and animals for use in a manner which will not cause ecological imbalance. Watershed management not only increases the production and income of the watershed community, but also mitigates droughts and floods and increases the life of downstream dam and reservoirs. Several organisations across the country have been working on rejuvenating ancient systems of water harvesting as an alternative to the dams. They have used several water saving methods to capture every trickle of water that has fallen on land, dug small pits and lakes and put in place simple watershed systems, built small earthen dams, constructed dykes, sand and limestone reservoirs and set up rooftop water-collecting units. This has recharged groundwater levels and even brought rivers back to life.
Answer 15.
The advantages and disadvantages of using solar cookers is as follows :
Advantages of using solar cooker:
- The use of solar cooker for cooking food saves precious fuels like coal, kerosene etc.
- The solar cooker does not produce any smoke due to which it does not pollute air.
- The food cooked in solar cooker does not lose its nutrients because food prepares at low temperature.
- It requires little maintenance.
Disadvantages of using solar cooker:
- The solar cooker cannot be used to cook food during night time, cloudy days and rainy days.
- The solar cooker cannot be used for baking and frying the food.
- The cost of setting up a solar cooker is quite high.
- The direction of reflector of solar cooker has to be changed from time to time to keep it facing the sun.
Answer 16.
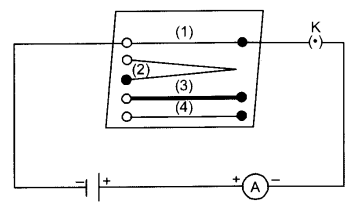
The figure above shows an electric circuit consisting of a cell, an ammeter, a nichrome wire and a plug key.
- It is observed that the ammeter reading decreases to one-half when the length of wire is doubled.
- The ammeter reading increases when a thicker wire of the same material and of the same length is used.
- If a wire of different material of the same area when used produces a change in ammeter reading. It is observed that the resistance of a conductor depends on its length, its area of cross-section and the nature of its material. Precise measurements have shown that the resistance of a uniform metallic conductor is directly proportional to its length (/) and inversely proportional to the area of cross-section (A)
So
Resistance, R ∝ l
R ∝ l/A
or R ∝ l/A or R=pl/A
Here p is the constant of proportionality and is called the electrical resistivity of the material of the conductor. The SI unit of resistivity is Ωm. It is a characteristic property of the material. Generally metals and alloys have low resistivity in the range of 10-8 Ωm to 10-6 Ωm. So they are good conductors of electricity. On the other hand non-metals like rubber and glass have high resistivity of the order 1012 to 1017 Ωm Thus, they are poor conductors of electricity.
Answer 17.
(a) As the concentration of hydrogen ions increases, the solution becomes more acidic.
(b) As the concentration of hydroxide ions increases, the solution becomes more basic.
(c) Vinegar is acidic in nature.
(d) Soap is basic in nature.
(e)
- pH = 9; Alkaline.
- pH = 4; Acidic.
- pH = 7: Neutral.
- pH = 1: Acidic,
- pH = 10: Alkaline,
- pH = 3 : Acidic.
OR
(a) Indicator is any substance that gives visible sign, usually by colour change, of the presence or absence of a chemical species. Two indicators are litmus and turmeric.
(b) Na2O + H2O → 2NaOH
NaOH in phenolphthalein shows pink colour and turns red litmus blue.
(c) NaOH + HCl (dil.) → NaCl + H2O
This is the neutralisation reaction where acid and base reacts to give salt and water.
Answer 18.
Ethanol (C2H5OH) is a commercially important hydrocarbon.
- It is a liquid at room temperature.
- It is commonly called alcohol and is an active ingredient of all alcoholic drinks.
- Being a good solvent, it is used in medicines like cough syrup, tonics and tincture iodine.
- Consumption of even a small amount of pure alcohol called absolute alcohol can be deadly.
- Consumption of alcoholic drinks is a social problem and it leads to many harmful effects.
Chemical properties of ethanol:
(i) Reaction with sodium: Alcohols react with sodium leading to the evolution of hydrogen. Sodium ethoxide is formed when sodium reacts with ethanol.
2Na + 2C2H5OH → 2C2H5ONa + H2
(ii) Reaction on heating: Heating ethanol at 443K with excess concentrated sulphuric acid results in the dehydration of ethanol to produce ethane.
C2H5OH \(\underrightarrow { hotconc.{ H }_{ 2 }S{ O }_{ 4 } } \) +C2H4 + H2O
As concentrated sulphuric acid removes water molecule from ethanol it is called a dehydrating agent.
Answer 19.
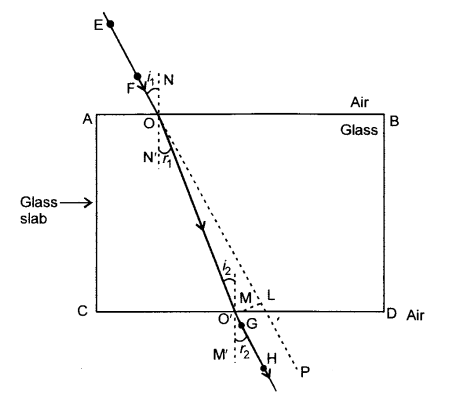
Place a rectangular glass slab on a white paper pinned firmly to a drawing board. Draw the outline of the slab neatly with a pencil and name it ABCD. Take 4 identical pins. Fix two pins E and F vertically one behind another such that the line joining them is inclined to side/edge AB. Look at these pins from the opposite edge and fix the two other pins G and H vertically in such a way that all four pins appear to be in a straight line. Now remove the slab and the pins. Join the positions E and F and extend the straight line to side AB. Let EF meet AB at O. Next join the positions of G and H and extend the line to meet side CD at O’. Now join O and O’ with a straight line. Extend EF towards point P beyond CD as a dotted line. This ray diagram helps us make the following observations:
- If NN’ is a perpendicular at O to side AB, angle EON is the angle of incidence.
- Angle N’OO’ is the angle of refraction.
- The above two angles are when the ray of light moves from air medium to glass medium.
- If MM’ is a perpendicular at O’ to side CD, angle OO’M is the angle of incidence and angle HO’M’ is the angle of refraction.
- The above two angles are when the ray of light moves from glass medium to air medium.
- EO is the incident ray, OO’ is the refracted ray and O’H is the emergent ray.
Refractive index is the ratio of the sine of the angle of incidence to the sine of the angle of refraction when a ray of light travels from one medium to another. This is always a constant.
Answer 20.
| Soaps | Detergents |
| (i) Soaps are sodium salts of long chain carboxylic acids. | Detergents are sodium salts of long chain benzene sulphonic acids. |
| (ii) The ionic group in soap is COONa+. | The ionic group in detergents is SO3–, Na+. |
| (iii) Soaps are not useful in hard water. | Detergents can be used even in hard water. |
| (iv) Soaps are biodegradable. | Detergents are not biodegradable. |
| (v) Soaps have relatively weak cleansing action. | Detergents have powerful cleansing action. |
Mechanism of the cleansing action of soaps : The dirt present on clothes is organic matter and is insoluble in water. Therefore, it cannot be moved by just washing with water. When soap dissolves in water, its hydrophobic ends attach themselves to the dirt and removes it from the cloth. Then the molecules arrange themselves in micelles form and trap the dirt in the centre of the cluster. These micelles remain suspended in the water. Thus, the dirt particles are easily removed by water.
Soaps do not lather with hard water : Soap is a sodium or potassium salt of long chain fatty acids. Hard water contains salts of calcium and magnesium. When soap is added to hard water, calcium and magnesium ions present in water displaces sodium or potassium ions present in soap forming insoluble substances called scum. Plenty of soap is wasted in this process.
Problems that arise due to the use of detergents instead of soap:
- As detergents are non-biodegradable they pollute the environment. When they add up in the soil they cause changes in the pH and make the soil infertile.
- Presence of detergents in the food chain causes health problems. Bio-accumulation of these substances causes serious health problems.
Answer 21.
Our environment refers to the various natural things around us both living and non-living. It includes the living things like plants and animals. It also includes the non-living things like air, water and the land. It has been seen that with industrial revolution and the progress in mankind’s standard of living the first thing to be affected is the environment. Mankind which is just a small part of this environment has played havoc on it in the last few decades. It is high time we realise that many of these resources are not going to last forever. An increasing human population with an increasing length of life is taxing the resources available. The only way out is for each one of us to adopt environment friendly measures in our daily lives. Here are some simple ways each one of us can prevent exploiting the environment.
- Let us use only as much as we need and not for our greed. Our houses can be small and compact made up of nature friendly material. We can have a natural cooling system with enough windows and some space for a little garden in the house. Natural cooling can minimise the use of air- conditioners and thereby reduce power consumption.
- Our homes must have facility to harvest rain water. It can be a simple process where we store the rain water and do not allow it to run away.
- If possible we can install solar panels for generating current and also cooking purpose. India being a country with plenty of sunshine, we must explore the use of solar energy wherever we can.
- The garbage we generate must be separated into biodegradable and non-biodegradable parts. The biodegradable garbage can be used to make compost which can be used for the plants. This helps reduce dependency on chemical fertilisers or manures.
- We must strictly adopt the three Rs in our daily lives so that we can minimise wastage and the pressure on our resources.
OR
Grass → Grasshopper → Frog → Snake → Eagle
The above sequence represents a food chain operating in our ecosystem. The series of organisms which eat and are eaten by others is called a food chain. Each level of the food chain is called a trophic level. Generally the autotrophs or producers are the first trophic level. They trap the solar energy and make it available for the heterotrophs or consumers. The herbivores or the primary consumers come at the second level, small carnivores or secondary consumers come at the third level and larger carnivores or tertiary consumers come at the fourth trophic level. Food forms the source of energy for each level of organism and helps it perform the various functions of life.
There is a flow of energy from one trophic level to another. Solar energy is trapped by the autotrophs during photosynthesis. This energy flows from one trophic level to the next. It has been found that only 1 / 10th of the energy is capable of moving to the next level. That is why as we move up the food chain we find fewer and fewer organisms. Also every food chain does not have more than three to four levels. The length and complexity of the food chains vary greatly. Each organism is generally eaten by two or more organisms which in turn are eaten by several others. So in any ecosystem, instead of a single straight food chain we can see many branching chains forming a food web.
SECTION-B
Answer 22.
(d) A and D
Answer 23.
CH3COOH + NaHCO3 → CH3COONa + CO2 + H2O
The gas evolved is C02. It is tested by passing it through lime water which turns milky in the presence of C02 due to the formation of CaC03.
Ca(OH)2 + CO2 → CaCO3 + H2O
Answer 24
(d) Forelimbs of a dog, frog and a crocodile.
Answer 25.
(d) To prove that light is necessary for photosynthesis.
Answer 26.
The required ray diagram is shown below:
Answer 27.
(c) Value of resistance changes,
(d) Resistor gets heated.
We hope the CBSE Sample Papers for Class 10 Science Paper 1 help you. If you have any query regarding CBSE Sample Papers for Class 10 Science Paper 1, drop a comment below and we will get back to you at the earliest.
